
Concept explainers
Interpretation:
Cyanide ion, carbonate ion, acetate ion can be considered as organic or not is to be determined.
Concept introduction:
Compounds can be classified as organic and inorganic material on the basis of the material from which they are derived. The organic compound is the class of compound that has carbon-hydrogen bond or carbon-carbon long chains in their structure. They are derived from plants, animals and living organisms.
Inorganic compounds are those compounds that consist of other elements than carbon or have no carbon-carbon bond in their structure. They are derived from minerals.
Answer to Problem 1E
Acetate ion
Explanation of Solution
Cyanide ion
Acetate ion
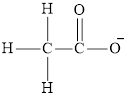
Acetate ion
Want to see more full solutions like this?
Chapter 21 Solutions
Bundle: Introductory Chemistry: An Active Learning Approach, 6th + LMS Integrated for OWLv2, 4 terms (24 months) Printed Access Card
- What is the full structure of an amine which is an isomer of ethylamine? What is the isomer called? Is it primary, secondary or tertiary?arrow_forwardA. COMPOSITIONHeat strongly a pinch of sugar on a broken crucible. Observe.Repeat the experiment using: a) a leaf (direct heating)b) a piece of plastic (direct heating)c) aluminum foil (direct heating)d) a pinch of starch (in a test tube)e) a pinch of sand. (in a test tube) Which substances are organic? What element is common to all organic compounds?arrow_forwardCompounds that generally have non-polar covalent bonds between atoms in the molecule would be classified as ________ compounds. - I am confused and not sure if the answer should only be organic? Or would it be both organic and inorganic.arrow_forward
- What are the carbon containing compounds that are not organic?arrow_forwardIn the compound bromomethane, does it matter which Hydrogen atom is removed and replaced with Bromine? Why or why not?arrow_forwardWhat is the attitude being portrayed by the scientist Friedrich Wohler in his contributions to organic chemistry?arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning





