
Concept explainers
PRACTICE PROBLEM
If we examine Table 21.1, we find that the methylphenols (cresols) are less acidic than phenol itself. For example,
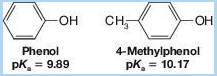
This behavior is characteristic of phenols bearing electron-releasing groups. Provide an explanation.
Interpretation:
Methyl phenols are less acidic than phenols, due to the presence of an electron releasing group on them. The statement is to be explained.
Concept introduction:
Phenols are compounds of benzene, bearing a hydroxyl group. They are alcohol derivatives but have higher acidities than alkyl alcohols.
Methyl phenols are phenol derivatives, in which a hydrogen atom attached to the carbon ring is replaced by a methyl
Answer to Problem 1PP
Solution: The
Explanation of Solution
According to table
Phenol is more acidic, as the
Want to see more full solutions like this?
Chapter 21 Solutions
Organic Chemistry (Looseleaf) (Custom Package)
Additional Science Textbook Solutions
Chemistry: Matter and Change
General, Organic, and Biological Chemistry (3rd Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Living by Chemistry
Living By Chemistry: First Edition Textbook
General Chemistry: Principles and Modern Applications (11th Edition)
- Could someone please help me with this practice problem? For each equilibrium below, circle the side that would be favored based on your understanding of factors affecting acid strength. In each case, briefly explain your choice. Thank you!arrow_forwardSIDE QUESTION (please reference image for actual question) Is it possibly reductive amination and hoffman elimination? Not sure how to get NH to be NH2 or if NH can be removed with hoffman?arrow_forwardSecond time asking, don't copy from previous answer, only typed answer pleasearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





