
(a)
Interpretation:
The number of
Concept Introduction:
There are three types of
(a)
Explanation of Solution
The given compound is shown here:
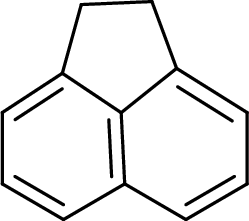
No. of double bonds:
So, the total number of
(b)
Interpretation:
The number of
Concept Introduction:
There are three types of
(b)
Explanation of Solution
The given compound is shown here:
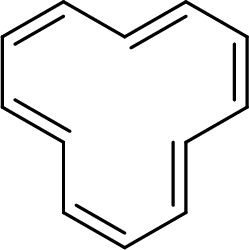
No. of double bonds:
So, the total number of
(c)
Interpretation:
The number of
Concept Introduction:
There are three types of
(c)
Explanation of Solution
The given compound is shown here:
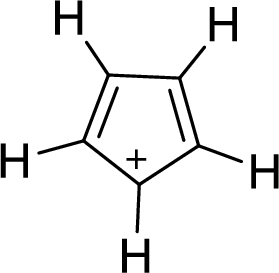
No. of double bonds:
So, the total number of
(d)
Interpretation:
The number of
Concept Introduction:
There are three types of
(d)
Explanation of Solution
The given compound is shown here:
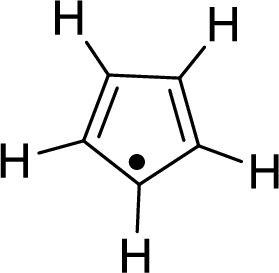
No. of double bonds:
So, the number of
There is an unpaired free-radical electron which is represented by a single dot. This unpaired free-radical electron will be in resonance with the
Therefore, the total number of
(e)
Interpretation:
The number of
Concept Introduction:
There are three types of
(e)
Explanation of Solution
The given compound is shown here:
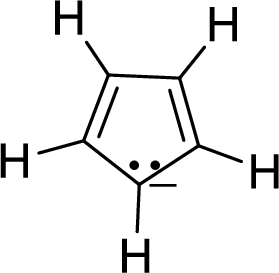
No. of double bonds:
So, the number of
There is a pair of electrons which is represented by two dots with a negative sign. This pair of electrons will be in resonance with the
Therefore, the total number of
(f)
Interpretation:
The number of
Concept Introduction:
There are three types of
(f)
Explanation of Solution
The given compound is shown here:
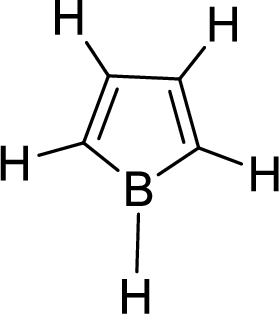
No. of double bonds:
So, the total number of
(g)
Interpretation:
The number of
Concept Introduction:
There are three types of
(g)
Explanation of Solution
The given compound is shown here:
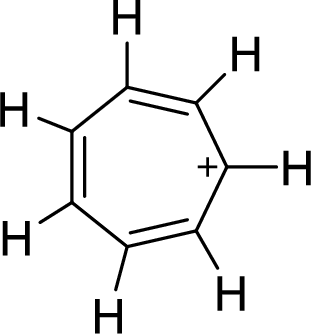
No. of double bonds:
So, the total number of
(h)
Interpretation:
The number of
Concept Introduction:
There are three types of
(h)
Explanation of Solution
The given compound is shown here:
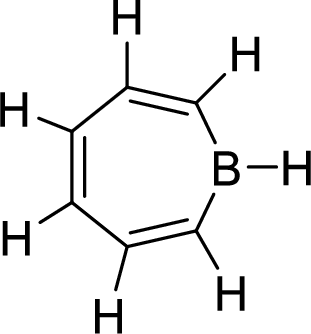
No. of double bonds:
So, the total number of
(i)
Interpretation:
The number of
Concept Introduction:
There are three types of
(i)
Explanation of Solution
The given compound is shown here:
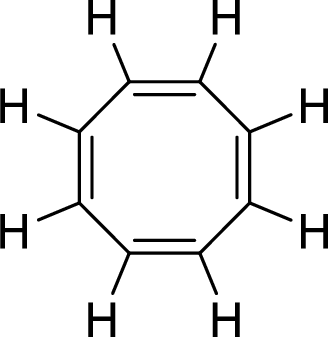
No. of double bonds:
So, the total number of
(j)
Interpretation:
The number of
Concept Introduction:
There are three types of
(j)
Explanation of Solution
The given compound is shown here:
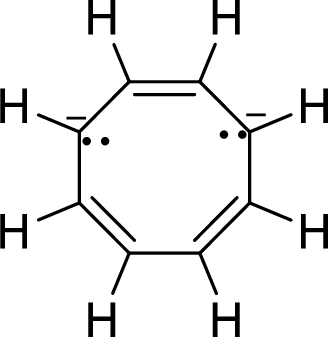
No. of double bonds:
So, the number of
There are two pairs of electrons and each pair is represented by two dots with a negative sign. These two pair of electrons will be in resonance with the
Therefore, the total number of
Want to see more full solutions like this?
Chapter 21 Solutions
ORG.CHEM:TXT+OWLV2+MINDTAP 6MTHS >BI<
- Identify hybridization in each Carbon atom of given molecules.arrow_forwardDraw the complete p molecular orbital diagram (energy diagram) for allyl anion and fill up the orbitals with the appropriate number of p electrons. As well as the cation please ( "draw the resonance structure before starting the energy diagram")arrow_forwardHow many p orbitals are present on the following molecule?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





