
Concept explainers
(a)
Interpretation:
The compound from given spectra is to be identified.
Concept Introduction:
In the IR spectroscopy, change in the dipole moment produces absorption of energy. All the functional group have different absorption frequency based on which they can be differentiated. For carbonyl
Answer to Problem 21.55AP
The structure of compound is shown below.
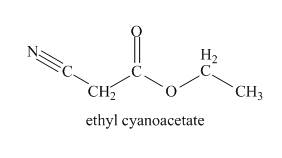
Explanation of Solution
The odd molecular mass of the compound indicates that it contains odd number of nitrogen atoms. Hydroxamate test is given by esters. So, compound contains ester group. The IR band at

Figure 1
The given compound is ethyl cyanoacetate.
(b)
Interpretation:
The compound from given spectra is to be identified.
Concept Introduction:
In the IR spectroscopy, change in the dipole moment produces absorption of energy. All the functional group have different absorption frequency based on which they can be differentiated. For carbonyl
Answer to Problem 21.55AP
The structure of given compound is shown below.
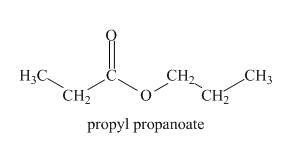
Explanation of Solution
The given compound gives IR absorption peak at

Figure 2
The given compound is propyl propanaote.
(c)
Interpretation:
The compound from given spectra is to be identified.
Concept Introduction:
In the IR spectroscopy, change in the dipole moment produces absorption of energy. All the functional group have different absorption frequency based on which they can be differentiated. For carbonyl
Answer to Problem 21.55AP
The structure of given compound is shown below.
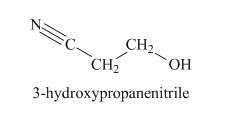
Explanation of Solution
The strong absorption peak in IR at

Figure 3
The given compound is
(d)
Interpretation:
The compound from given spectra is to be identified.
Concept Introduction:
In the IR spectroscopy, change in the dipole moment produces absorption of energy. All the functional group have different absorption frequency based on which they can be differentiated. For carbonyl
Answer to Problem 21.55AP
The structure of given compound is shown below.
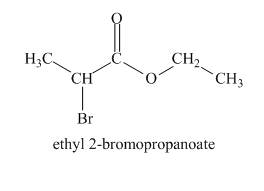
Explanation of Solution
The two molecular ion peaks at

Figure 4
The given compound is
(e)
Interpretation:
The compound from given spectra is to be identified.
Concept Introduction:
In the IR spectroscopy, change in the dipole moment produces absorption of energy. All the functional group have different absorption frequency based on which they can be differentiated. For carbonyl
Answer to Problem 21.55AP
The structure of given compound is shown below.
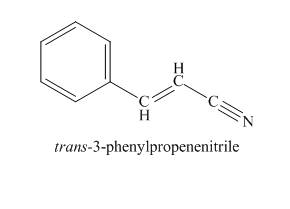
Explanation of Solution
The compound has odd molecular mass which indicates the presence of nitrogen atom. The IR absorption peak at

Figure 5
The given compound is
(f)
Interpretation:
The compound from given spectra is to be identified.
Concept Introduction:
In the IR spectroscopy, change in the dipole moment produces absorption of energy. All the functional group have different absorption frequency based on which they can be differentiated. For carbonyl
Answer to Problem 21.55AP
The structure of given compound is shown below.
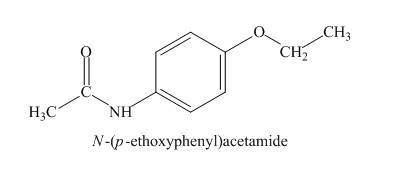
Explanation of Solution
The absorption in IR at ![]() indicates the presence of amide group and at
indicates the presence of amide group and at
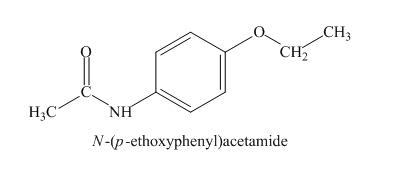
Figure 6
The given compound is
(g)
Interpretation:
The compound from given spectra is to be identified.
Concept Introduction:
In the IR spectroscopy, change in the dipole moment produces absorption of energy. All the functional group have different absorption frequency based on which they can be differentiated. For carbonyl
Answer to Problem 21.55AP
The structure of given compound is shown below.
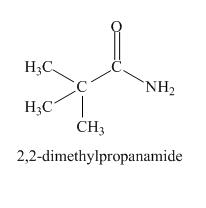
Explanation of Solution
The odd molecular mass of the compound indicates it contains nitrogen atom. The IR absorption peak at

Figure 7
The given compound is
Want to see more full solutions like this?
Chapter 21 Solutions
ORGANIC CHEMISTRY (LL)+ SAPLING ACC >BI
- Answer the following questions on using IR spectroscopy to characterize the products in the synthesis ofluminol.a. Can IR be used to determine if the synthesis of the hydrazide was successful? Explain youranswer.b. IR cannot be used to determine the success of the reduction of the nitro group into an amine in Part II.Explain why not.arrow_forwardHow could 1H NMR spectroscopy be used to distinguish among isomers A, B, and C?arrow_forwardPropose a structure for D (molecular formula C9H9ClO2) consistent with the given spectroscopic data. 13C NMR signals at 30, 36, 128, 130, 133, 139, and 179 ppmarrow_forward
- An unkown compound, which is insoluable in water but soluble in 10% aqueous sodium bicarbonate, yielded the given 1H NMR spectrum. What is the compound?arrow_forwardA chiral compound Y has a strong absorption at 2970−2840 cm−1 in its IR spectrum and gives the following mass spectrum. Propose a structure for Y.arrow_forwardCompound K, molecular formula C6H14O, readily undergoes acid-catalyzed dehydration when warmed with phosphoric acid to give compound L, molecular formula C6H12, as the major organic product. The 1H-NMR spectrum of compound K shows signals at d 0.90 (t, 6H), 1.12 (s, 3H), 1.38 (s, 1H), and 1.48 (q, 4H). The 13C-NMR spectrum of compound K shows signals at d 72.98, 33.72, 25.85, and 8.16. Deduce the structural formulas of compounds K and L.arrow_forward
- These are the 1H RMN and IR spectrums of an A compound with formula C10H13NO2. When A reacts with NaOH in water and heat, B compound is formed, with formula C10H11NO. What are the structures of A and B?arrow_forwardCompound P has molecular formula C5H9C102. Deduce the structure of P from its 1H and 13C NMR spectra.arrow_forwardCompound A is a hydrocarbon with a molar mass of 96g/mol, with the given C13 spectral data. When compound A reacts with BH3 followed by the treatment with basic H2O2 it is converted to compound B. Propose structures for A and B, explain your analysis.Compound A- Proton decoupled C NMR: 26.8, 28.7, 35.7, 106.9, 149.7 δ.DEPT-90: No peak.DEPT-135: No positive peaks; negative peaks at 26.8, 28.7, 35.7, 106.9 δ.Compound B- Proton decoupled C NMR: 26.1, 26.9, 29.9, 40.5, 68.2 δ.DEPT-90: 40.5 δ.DEPT-135: positive peak at 40.5 δ; negative peaks at 26.1, 26.9, 29.9, 68.2 δarrow_forward
- compound with the molecular formula C7H9N exhibits IR bands at 3450 cm-1 (medium, doublet), and 855 cm-1 (strong) and shows the following major mass spectral signals (m/z): 106 (base); 107 (M+, about 70% of base), 91 (40% of base), and 77 (about 20% of base). Deduce a reasonable structure from this data.arrow_forwardReaction of butanenitrile (CH3CH2CH2CN) with methylmagnesium bromide (CH3MgBr), followed by treatment with aqueous acid, forms compound G. G has a molecular ion in its mass spectrum at m/z = 86 and a base peak at m/z = 43. G exhibits a strong absorption in its IR spectrum at 1721 cm−1 and has the 1H NMR spectrum given below. What is the structure of G? We will learn about the details of this reaction in Chapter 22.arrow_forwardReaction of pentanoyl chloride (CH3CH2CH2CH2COCl) with lithium dimethyl cuprate [LiCu(CH3)2] forms a compound J that has a molecular ion in its mass spectrum at 100, as well as fragments at m/z = 85, 57, and 43 (base). The IR spectrum of J has strong peaks at 2962 and 1718 cm-1. Propose a structure for J.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
