
Concept explainers
(a)
Interpretation:
Electronic configuration of the following metals has to be written –
Concept Introduction:
Electronic configuration of an atom represents the arrangement of electrons in various energy levels. The electrons are arranged in increasing order of energy levels according to Aufbau principle. It is pictorially represented as –
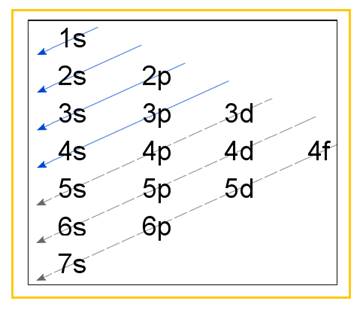
Figure 1
The terms
(a)
Answer to Problem 22E
Electronic configuration of
Explanation of Solution
The above electronic configuration corresponds to that of Argon till
(b)
Interpretation:
Electronic configuration of the following metals has to be written –
Concept Introduction:
Electronic configuration of an atom represents the arrangement of electrons in various energy levels. The electrons are arranged in increasing order of energy levels according to Aufbau principle. It is pictorially represented as –

Figure 1
The terms
(b)
Answer to Problem 22E
Electronic configuration of
Explanation of Solution
Atomic number of Cadmium is
The above electronic configuration corresponds to that of Argon till
(c)
Interpretation:
Electronic configuration of the following metals has to be written –
Concept Introduction:
Electronic configuration of an atom represents the arrangement of electrons in various energy levels. The electrons are arranged in increasing order of energy levels according to Aufbau principle. It is pictorially represented as –
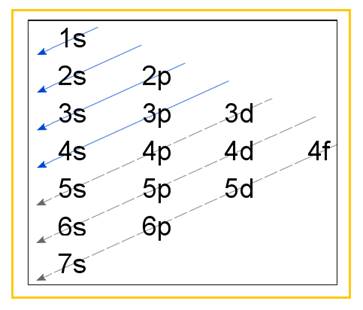
Figure 1
The terms
(c)
Answer to Problem 22E
Electronic configuration of
Explanation of Solution
Atomic number of Zirconium is
The electronic configuration of
The above electronic configurations correspond to that of Argon till
(d)
Interpretation:
Electronic configuration of the following metals has to be written –
Concept Introduction:
Electronic configuration of an atom represents the arrangement of electrons in various energy levels. The electrons are arranged in increasing order of energy levels according to Aufbau principle. It is pictorially represented as –
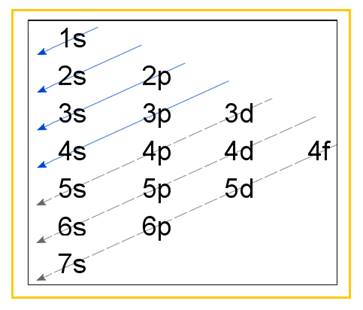
Figure 1
The terms
(d)
Answer to Problem 22E
Electronic configuration of
Explanation of Solution
Atomic number of Osmium is
Os2+ and Os3+ are formed when Osmium loses two electrons and three electrons respectively. Accordingly the electronic configuration of Os2+ is –
The above electronic configuration corresponds to that of Xenon till
The electronic configuration of
The above electronic configuration corresponds to that of Xenon till
Want to see more full solutions like this?
Chapter 21 Solutions
EBK CHEMISTRY
- Give the electron distribution in low-spin and/or high-spin complexes of (a) Ru4+ (b) Pt2+arrow_forwardThe spectrochemical series:a. lists metals ions in order of their oxidation state.b. lists metals ions in order by their color.c. lists ligands in order of their tendency to split d orbitals.d. is used to determine the oxidation state of a central metal atom.e. is used to calculate the crystal field splitting energy of complex ions. deltaS will be positive for the reaction a. 2H2 (g) + O2 (g) → 2H2O (g)b. 2NO2 (g) → N2O4 (g)c. NaF2 (aq) → Na+(aq) + 2F¯(aq)d. 2Hg (l) + O2 (g) → 2HgO (s)e. CO2 (g) → CO2 (s)arrow_forwardWrite the formula for diaquadibromoplatinum (II). C [Pt(H2O)2Br2]2 C [Pt(H2O)2]Br2*1 C [Pt(H2O)2]Br2 C [Pt(H2O)2]Br22 C (Pt(H2O)2Br2] C PE(H,O),Bra]*1arrow_forward
- What is the percentage of oxalate in each of the following compounds? a. K3[Cr(C2O4)3]3H2Ob. K2[Cu(C2O4)2]2H2O c. K3[Fe(C2O4)3]3H2Od. K3[Al(C2O4)3]3H2Oarrow_forwardWhy dp transition elements show variable oxidation states? In 3d series (Sc to Zn), which element shows the maximum number of oxidation states and why?arrow_forwardWhy do ligands cause the d orbitals of a transition metal ion to cease being degenerate? A. New molecular orbitals are formed via atomic orbital overlapt B. There is repulsion between the ligands and the d orbitals. C. They destabilize the ion nucleus D. Donation of electrons to the metal ion results in redox chemistryarrow_forward
- 1. What mass of solid Lanthanum (III) oxalate nonahydrate { La2 ( C2O4 )3 ∙ 9 H2O } can be obtained from 650 mL of 0.0170 M aqueous solution of LaCl3 by adding a stoichiometric amount of sodium oxalate?arrow_forwardThe oxidation of 22 mL of a solution containing Fe2+ requires 27 mL of 0.0345 M K2Cr2O7 in acidic solution. Balance the following equation. Do NOT include the states of each species. a. Cr2O72− + Fe2+ + H+ → Cr3+ + Fe3+ b. Calculate the molar concentration of Fe2+.arrow_forwardHow many unpaired electrons are in the following complex ions? a. Ru(NH3)62+ (low-spin case) b. Ni(H2O)62+ c. V(en)33+arrow_forward
- What is the coordination number of the central ion? [Cr(H2O)6]*3 ? O a. 3 O b. 6 с. 1 O d. 2arrow_forwardSulfide (S) salts are notoriously insoluble in aqueous solution. a. Calculate the molar solubility of nickel(II) sulfide in water. K„(NiS) = 3 x 10-16 b. Nickel(11) ions form a complex ion in the presence of ammonia with a formation constant (K) of 2.0 x 10*: Ni* + 6 NH, = [Ni(NH,),12+. Calculate the molar solubility of NiS in 3.0 M NH3. c. Explain any differences between the answers to parts a and b.arrow_forwardWrite electron configurations for each of the following . a. Ti, Ti2+ Ti4+ b. Re,Re2+, Re3+ c. Ir, Ir2+, Ir3+arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER





