
ORGANIC CHEMISTRY (LL) W/WILEYPLUS NEXT
12th Edition
ISBN: 9781119664635
Author: Solomons
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21, Problem 28P
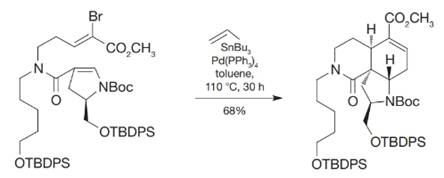
During the course of the following Stille coupling, the product shown was formed. How did this result come about?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The following reaction resulted in the product shown below, but not the other product shown. Using the reaction mechanisms and chemical structures, explain why their products was obtained. The product was obtained as a racemic mixture.
Suggest a detailed mechanism for each of the reactions below. Explain why the originating products were formed.
Provide a detailed mechanism for this reaction
Chapter 21 Solutions
ORGANIC CHEMISTRY (LL) W/WILEYPLUS NEXT
Ch. 21 - PRACTICE PROBLEM 21.1
For each of the following...Ch. 21 - Prob. 2PPCh. 21 - PRACTICE PROBLEM 21.3 What product would you...Ch. 21 - Prob. 4PPCh. 21 - PRACTICE PROBLEM 21.5 What is the product of the...Ch. 21 - Prob. 6PPCh. 21 - Prob. 7PPCh. 21 - Prob. 8PPCh. 21 - Prob. 9PPCh. 21 - Prob. 10PP
Ch. 21 - Prob. 11PPCh. 21 - Practice Problem 21.12 What products would form...Ch. 21 - Prob. 13PCh. 21 - Prob. 14PCh. 21 - How would you synthesize each of the following...Ch. 21 - Prob. 16PCh. 21 - Predict the product(s) for each of the following...Ch. 21 - Prob. 18PCh. 21 - Prob. 19PCh. 21 - Prob. 20PCh. 21 - Prob. 21PCh. 21 - 21.22 Write a mechanism that can account for the...Ch. 21 - Prob. 23PCh. 21 - Prob. 24PCh. 21 - Prob. 25PCh. 21 - 21.26 In 1985, T. Katz (Columbia University)...Ch. 21 - When the following molecule was exposed to the...Ch. 21 - During the course of the following Stille...Ch. 21 - 1. In “The Chemistry of... Complex Cross...Ch. 21 - Prob. 2LGP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
37. Balance each redox reaction occurring in acidic aqueous solution.
a. K(s) + Cr3+(aq) → Cr(s) + K+(aq)
b. Al...
Chemistry: A Molecular Approach
For the generic equilibrium HA(aq) ⇌ H + (aq) + A- (aq), which of these statements is true?
The equilibrium con...
Chemistry: The Central Science (14th Edition)
Protein is to be described. Concept Introduction : Our body is made of many types of molecules. There are body ...
Living By Chemistry: First Edition Textbook
10.71 Identify each of the following as an acid or a base: (10.1)
H2SO4
RbOH
Ca(OH)2
HI
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Give a molecular orbital description for each of the following: a. 1,3-pentadiene b. 1,4-pentadiene c. 1,3,5-he...
Organic Chemistry (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Provide a detailed mechanism for the following reactionarrow_forwardIdentify compounds G,H,E in the following reaction sequences.arrow_forwardWhen ethylbenzene reacts with bromine in a free radical halogenation, a small amount ofproduct that contains no bromine is formed. Show the mechanism of how the product isformed and the product. Explain why only a small amount of that product is formed.arrow_forward
- Identify the leaving group for a potential elimination of the following compounds. Compare the leaving group activities for A and K. Which of the following compounds cannot be subjected to elimination? Explain.arrow_forwardGive the mechanism and product of the following reactionarrow_forwardWhat is the major product structure? Provide the detailed mechanism of this reaction pleasearrow_forward
- Organometallic compounds act as nucleophiles under very specific conditions, and can generate alcohols, alkanes, among others. Describe in detail what these reaction conditions are so that organometallics can perform the addition reaction to carbonyl.arrow_forwardThe following reaction is Friedel-Crafts alkylation. Considering the mechanism for Friedel-Crafts alkylation, how would you explain the formation of this particular product? Either describe or upload image of the mechanism. AICI,arrow_forwardWhat is the mechanism (in detail) of the following reaction?arrow_forward
- Provide the complete mechanism in the following reactions.arrow_forwarddetermine which is the Hoffman elimination product from the two and provide the mechanism belowarrow_forwardProvide the mechanism and the structure of the products A and B in the given reactions. Explain the advantage of one over the other.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License