
Interpretation:
The equation for the reaction between propanoic acid and ethanol is to be written and the ester formed in this reaction is to be named.
Concept introduction:
Carboxylic acid reacts with alcohol to form an ester. This reaction is known as an esterification reaction or condensation reaction. In this reaction, a water molecule is lost. The general structure of an ester is as follows:
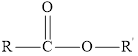
Here, R and
The steps to write the IUPAC name of an ester are as follows:
Step 1: Identify the parent chain. The alkyl group from the parent acid represents the parent chain.
Step 2: Number the carbon atoms. Since the parent chain is derived from carboxylic acid, therefore; the name of the parent acid is alkanoic acid. Drop the letters
Step 3: Number the carbon atoms of the alcohol part from the carbon which is directly bonded to the oxygen atom. The alcohol part is named as ‘alkyl’.
Step 4: In the case of ester, an alkyl group of the alcohol is written first followed by the anion of carboxylic acid. The format to name an ester is as follows:
Want to see the full answer?
Check out a sample textbook solution
Chapter 21 Solutions
Introductory Chemistry: An Active Learning Approach
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





