
Chemistry
9th Edition
ISBN: 9781133611097
Author: Steven S. Zumdahl
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21, Problem 90CP
Consider the pseudo-octahedral complex ion of Cr3+, where A and B represent ligands.
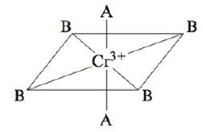
Ligand A produces a stronger crystal field than ligand B. Draw an appropriate crystal field diagram for this complex ion (assume the A ligands are on the z-axis).
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 21 Solutions
Chemistry
Ch. 21 - What two first-row transition metals have...Ch. 21 - Prob. 2RQCh. 21 - Prob. 3RQCh. 21 - Prob. 4RQCh. 21 - Prob. 5RQCh. 21 - Prob. 6RQCh. 21 - Prob. 7RQCh. 21 - Prob. 8RQCh. 21 - Prob. 9RQCh. 21 - Prob. 10RQ
Ch. 21 - Prob. 1ALQCh. 21 - Prob. 2ALQCh. 21 - Prob. 3ALQCh. 21 - Prob. 4ALQCh. 21 - Prob. 5QCh. 21 - Four different octahedral chromium coordination...Ch. 21 - Prob. 7QCh. 21 - Prob. 8QCh. 21 - Prob. 9QCh. 21 - Prob. 10QCh. 21 - Prob. 11QCh. 21 - Prob. 12QCh. 21 - Prob. 13QCh. 21 - Prob. 14QCh. 21 - Which of the following ligands are capable of...Ch. 21 - Prob. 16QCh. 21 - Prob. 17QCh. 21 - Prob. 18QCh. 21 - Prob. 19QCh. 21 - Prob. 20QCh. 21 - Prob. 21ECh. 21 - Prob. 22ECh. 21 - Prob. 23ECh. 21 - Prob. 24ECh. 21 - Prob. 25ECh. 21 - Prob. 26ECh. 21 - Prob. 27ECh. 21 - Prob. 28ECh. 21 - Prob. 29ECh. 21 - When an aqueous solution of KCN is added to a...Ch. 21 - Prob. 31ECh. 21 - A coordination compound of cobalt(III) contains...Ch. 21 - Prob. 33ECh. 21 - Prob. 34ECh. 21 - Prob. 35ECh. 21 - Prob. 36ECh. 21 - Prob. 37ECh. 21 - Give formulas for the following complex ions. a....Ch. 21 - Prob. 39ECh. 21 - Prob. 40ECh. 21 - Prob. 41ECh. 21 - Amino acids can act as ligands toward transition...Ch. 21 - Prob. 43ECh. 21 - Prob. 44ECh. 21 - Prob. 45ECh. 21 - Prob. 46ECh. 21 - Prob. 47ECh. 21 - Prob. 48ECh. 21 - Prob. 49ECh. 21 - Prob. 50ECh. 21 - Prob. 51ECh. 21 - Prob. 52ECh. 21 - The CrF64 ion is known to have four unpaired...Ch. 21 - Prob. 54ECh. 21 - Prob. 55ECh. 21 - The complex ion Fe(CN)63 is paramagnetic with one...Ch. 21 - Prob. 57ECh. 21 - Prob. 58ECh. 21 - Prob. 59ECh. 21 - Prob. 60ECh. 21 - The wavelength of absorbed electromagnetic...Ch. 21 - The complex ion NiCL42 has two unpaired electrons,...Ch. 21 - How many unpaired electrons are present in the...Ch. 21 - The complex ion PdCl42is diamagnetic. Propose a...Ch. 21 - Prob. 65ECh. 21 - Prob. 66ECh. 21 - Prob. 67ECh. 21 - Prob. 68ECh. 21 - Silver is sometimes found in nature as large...Ch. 21 - Prob. 70ECh. 21 - Prob. 71AECh. 21 - The compound cisplatin, Pt(NH3)2Cl2, has been...Ch. 21 - Prob. 73AECh. 21 - Prob. 74AECh. 21 - Prob. 75AECh. 21 - Prob. 76AECh. 21 - Prob. 77AECh. 21 - Name the following coordination compounds. a....Ch. 21 - Prob. 79AECh. 21 - Prob. 80AECh. 21 - Prob. 81AECh. 21 - Prob. 82AECh. 21 - Prob. 83CWPCh. 21 - Which of the following molecules exhibit(s)...Ch. 21 - Prob. 85CWPCh. 21 - The following table indicates the number of...Ch. 21 - Prob. 87CWPCh. 21 - Which of the following statement(s) is( are) true?...Ch. 21 - Consider the following complex ion, where A and B...Ch. 21 - Consider the pseudo-octahedral complex ion of...Ch. 21 - Prob. 91CPCh. 21 - Prob. 92CPCh. 21 - Chelating ligands often form more stable complex...Ch. 21 - Prob. 94CPCh. 21 - Prob. 95CPCh. 21 - Prob. 96CPCh. 21 - Sketch and explain the most likely crystal field...Ch. 21 - The ferrate ion, FeO42, is such a powerful...Ch. 21 - Ammonia and potassium iodide solutions are added...Ch. 21 - a. In the absorption spectrum of the complex ion...Ch. 21 - There are three salts that contain complex ions of...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
The Bohr Model of the atom and Atomic Emission Spectra: Atomic Structure tutorial | Crash Chemistry; Author: Crash Chemistry Academy;https://www.youtube.com/watch?v=apuWi_Fbtys;License: Standard YouTube License, CC-BY