
Concept explainers
Practice Problem ATTEMPT
ATTEMPT
Write empirical formulas for the following molecules: (a) caffeine
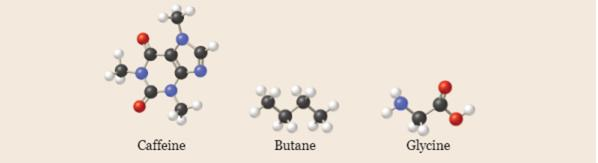
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
CHEMISTRY(LOOSELEAF W/CODE)- CUSTOM
- Practice Exercise 2Write the empirical formula for the compound formed by(a) Na+ and PO43-, (b) Zn2+ and SO42-, (c) Fe3+ and CO32-.arrow_forwardPractice Exercise 2Give the empirical formula for decaborane, whose molecularformula is B10H14arrow_forwardDirection: Find the molecular formula of the followingcompounds 7. A compound contains 62.0% C, 10.4% H, and27.5% O. Determine the empirical formula forthis compound. After analysis, it was found thatthe compound’s molar mass is 58.1g. What isthe compound’s molecular formula?8. Glucose, one of the main sources of energyused by living organisms, has a molecular massof 180.2g. Chemical analysis shows that glucoseis 40.0% C, 6.71% H and 53.3% O. Determineglucose’s molecular formula.9. A class of compounds called sodiummetaphosphates were used as additives todetergents to improve cleaning ability. One ofthem has a molecular mass of 612g. Analysisshows the composition to be 22.5% Na, 30.4% P,and 47.1% O. Determine the molecular formulaof this compound.10. Find the molecular formula for a compound thathas a molecular mass of 92g.mole. The %composition of the compound is 30.4% N, and69.6% O.arrow_forward
- Which of the following is not an empirical formula C2H6 C2H CH3 CHarrow_forwardA compound is 57.14% C, 6.16% H, 9.52% N, and 27.18% O. The molar mass is 588.6 g/mol. What is the molecular formula? Group of answer choices C14H18N5O2 C28H36N4O10 C7H9NO3 C14H18N2O5 C7H9NO5arrow_forwardMSG is monosodium glutamate, a flavor enhancer added to many foods. Monosodium glutamate was found to contain 35.51% C, 5.15% H, 37.85% O and 13.6% Na, and 8.26% N. It has a molar mass of 169 g/mol. What is the molecular formula of monosodium glutamate? Please show workarrow_forward
- What is the molecular formula of a compound with the empirical formula CH2O and a molar mass of 60.0 g? Group of answer choices C2H4O2 CH2O C2H2O2 C2H4O4arrow_forwardPractice Exercise 1Tetracarbon dioxide is an unstable oxide of carbon with thefollowing molecular structure:What are the molecular and empirical formulas of thissubstance? (a) C2O2, CO2 (b) C4O, CO (c) CO2, CO2(d) C4O2, C2O (e) C2O, CO2arrow_forwardWhat is the empirical formula of a compound that is found to be 61.4% vanadium and the rest is oxygen? Question 15 options: V3O5 VO2 VO V2O3 VO3arrow_forward
- 5, A mineral sample is obtained from a region of the country that has high arsenic contamination. An elemental analysis yields the following elemental composition: Element Atomic Weight (g/mol) Percent Composition Cu 63.546 30.2% Zn 65.409 15.5% As 74.9216 23.7% S 32.065 30.5% What is the empirical formula of this mineral? Group of answer choices A, Cu6Zn3As4S12 B, Cu5Zn3As4S12 C, Cu6Zn3As4S10 D, Cu6Zn3As3S12 Cu3Zn6As4S12arrow_forwardCalculate the number of moles of carbon atoms present in eachof the followingsamples.l type="a">1.271 g of ethanol,i>3.982 g of 1 ,4-dichlorobenzene,i>0.4438 g of carbon suboxide,i>2.9 10 g of methylene chloride,check_circle Expert SolutionInterpretation Introduction(a)Interpretation:Find launchC2H5OHC6H4Cl2C3O2CH2Cl2arrow_forward10647Ag → 10646Pd + ? a) it is n (mass # : 1; charge:0) b) no right answer c) it is Br (mass # : 76; protons: 35) d) it is C (mass #: 14; protons 6) e) it is e ( mass #: 0 ; charge +1)arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

