
Concept explainers
a)
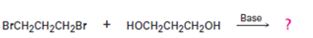
Interpretation:
The structure of the step-growth polymer expected to be obtained from the reaction given is to be shown.
Concept introduction:
In step growth
To draw:
The structure of the step-growth polymer expected to be obtained from the reaction given.
b)
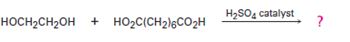
Interpretation:
The structure of the step-growth polymer expected to be obtained from the reaction given is to be given.
Concept introduction:
In step growth polymers each bond is formed independently in a discrete step. In the formation of these step growth polymers, n moles of one component react with n moles of other component to form a polymer with n repeating units eliminating 2n moles of small molecules.
To draw:
The structure of the step-growth polymer expected to be obtained from the reaction given.
c)
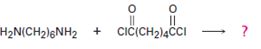
Interpretation:
The structure of the step-growth polymer expected to be obtained from the reaction given is to be given.
Concept introduction:
In step growth polymers each bond is formed independently in a discrete step. In the formation of these step growth polymers, n moles of one component react with n moles of other component to form a polymer with n repeating units eliminating 2n moles of small molecules.
To draw:
The structure of the step-growth polymer expected to be obtained from the reaction given.
Want to see the full answer?
Check out a sample textbook solution
Chapter 21 Solutions
Organic Chemistry
- Draw short segments of the polymers obtained from the following monomers. indicate whether the polymerization is a chain-growth or a step-growth polymerization.arrow_forwardDraw short segments of the polymers obtained from the following monomers. In each case, indicate whether the polymerization is a chain-growth or a step-growth polymerization.arrow_forwardDraw the structure of the polymer that results from anionic polymerization of p-trichloromethylstyrene (CCl3C6H4CH =CH2) using ethylene oxide as the electrophile to terminate the chain.arrow_forward
- I keep thinking the plastic sulfur is S8 or an Sx polymer. I'm leaning more towards the polymer of sulfur since when you heat the sulfur, it breaks its cyclic bond and makes these long chains but I'm completely confused.arrow_forwardLinear polymers are more water-soluble than branched ones. True Falsearrow_forwardIn most cases, end groups contribute insignificant to the physical/chemical properties of polymers. Among the types of polymers that we learned , which types of polymers' end group can contribute relatively largely?arrow_forward
- Give a schematic drawing of a linear, branched and cross-linked polymer, as well as an isotactic, atactic and syndiotactic polymer.arrow_forwardPredict the product of the following polymerization reactions. (Show only one repeating unit of the polymer in each reaction.)arrow_forwardDraw a structural formula of the polymer resulting from base-catalyzed polymerization of each compound. Would you expect the polymers to be optically active? (S)-(+)-lactide is the dilactone formed from two molecules of (S)-(+)-lactic acid.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning




