
Concept explainers
(a)
Interpretation:
Given compounds has to be arranged in their decreasing order of reactivity towards electrophilic
(a)
Explanation of Solution
Given compounds,
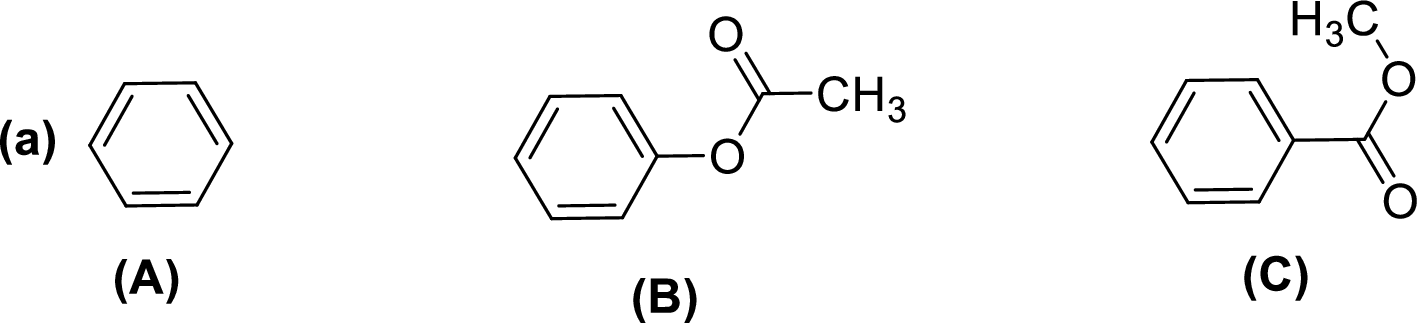
General trend of compounds reactivity in electrophilic aromatic substitution for a set of compounds having similar structure is given by,
So, electron donating substituents will increases the reactivity, while electron withdrawing substituents will decreases the reactivity.
Therefore, the decreasing order of reactivity towards electrophilic aromatic substitution of the given compounds are given below,
(b)
Interpretation:
Given compounds has to be arranged in their decreasing order of reactivity towards electrophilic aromatic substitution.
(b)
Explanation of Solution
Given compounds,
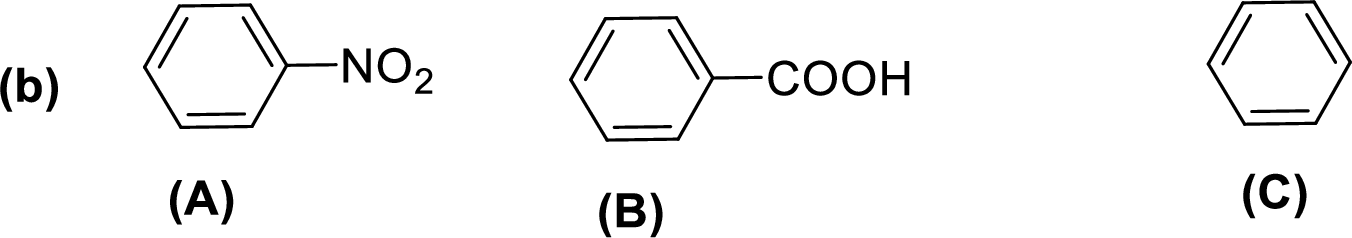
General trend of compounds reactivity in electrophilic aromatic substitution for a set of compounds having similar structure is given by,
So, electron donating substituents will increases the reactivity, while electron withdrawing substituents will decreases the reactivity.
Therefore, the decreasing order of reactivity towards electrophilic aromatic substitution of the given compounds are given below,
(c)
Interpretation:
Given compounds has to be arranged in their decreasing order of reactivity towards electrophilic aromatic substitution.
(c)
Explanation of Solution
Given compounds,
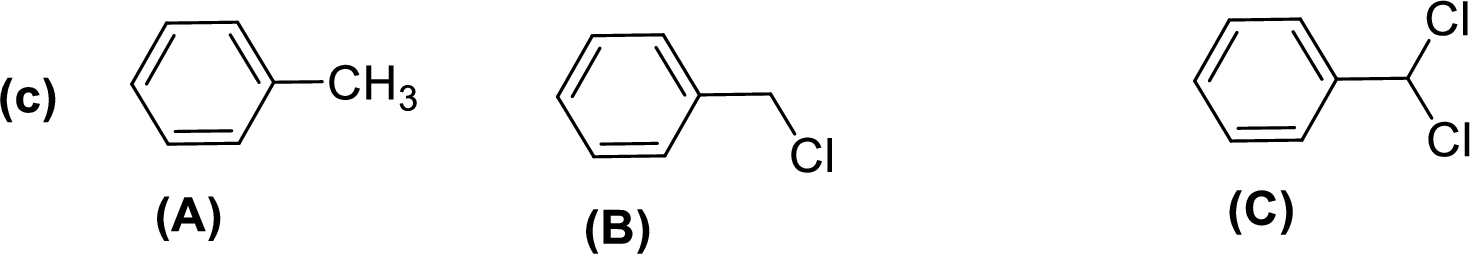
General trend of compounds reactivity in electrophilic aromatic substitution for a set of compounds having similar structure is given by,
So, electron donating substituents will increases the reactivity, while electron withdrawing substituents will decreases the reactivity.
Therefore, the decreasing order of reactivity towards electrophilic aromatic substitution of the given compounds are given below,
(d)
Interpretation:
Given compounds has to be arranged in their decreasing order of reactivity towards electrophilic aromatic substitution.
(d)
Explanation of Solution
Given compounds,
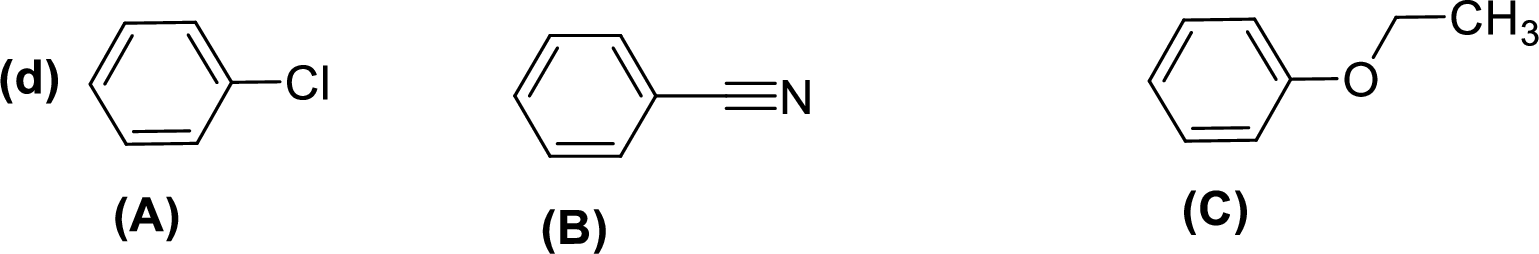
General trend of compounds reactivity in electrophilic aromatic substitution for a set of compounds having similar structure is given by,
So, electron donating substituents will increases the reactivity, while electron withdrawing substituents will decreases the reactivity.
Therefore, the decreasing order of reactivity towards electrophilic aromatic substitution of the given compounds are given below,
(e)
Interpretation:
Given compounds has to be arranged in their decreasing order of reactivity towards electrophilic aromatic substitution.
(e)
Explanation of Solution
Given compounds,
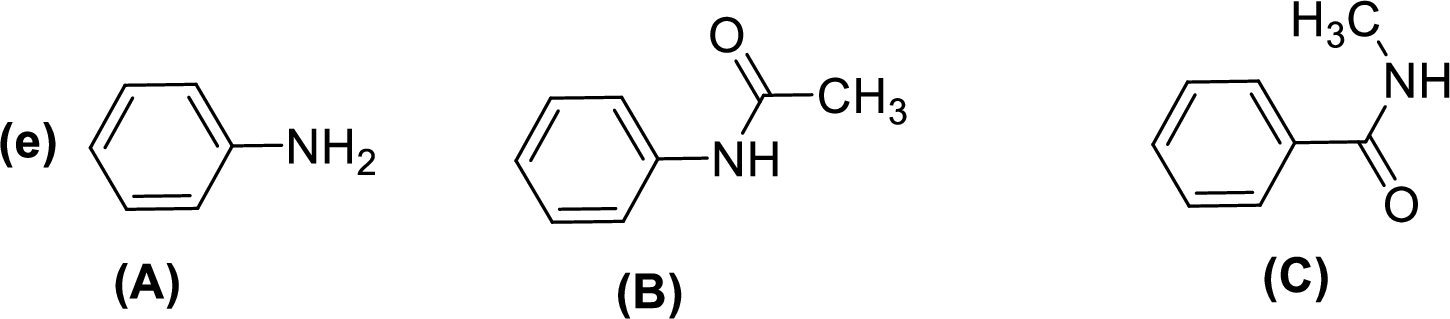
General trend of compounds reactivity in electrophilic aromatic substitution for a set of compounds having similar structure is given by,
So, electron donating substituents will increases the reactivity, while electron withdrawing substituents will decreases the reactivity.
Therefore, the decreasing order of reactivity towards electrophilic aromatic substitution of the given compounds are given below,
Want to see more full solutions like this?
Chapter 22 Solutions
ORG.CHEM:TXT+OWLV2+MINDTAP 6MTHS >BI<
- Rank the following groups in order of their ability to activate an aromatic compound to electrophilic aromatic substitution.arrow_forwardThe following molecule contain two aromatic rings.Which ring in each undergoes electrophilic aromatic substitution more readily? Draw the major product formed on nitration.arrow_forwardThe molecule below can be synthesized via two pathways using the SN2 reaction throughthe formation of the C-S bond. For each retrosynthetic pathway, draw the nucleophile andelectrophile and choose with pathway would be more efficient and explain whyarrow_forward
- Identify the ring which is more reactive towards electrophilic aromatic substitution.arrow_forwardQuinolines and isoquinolines which has a greater electrophilic aromatic substitution reactivityarrow_forwardarrange the following carbonyls in descending order according to their reactivityarrow_forward
- Propose an efficient synthesis of (a) and (b) of the following transformationsarrow_forwardPhenols have lower pKa values than other alcohols due to the resonance stabilization of the conjugate base. Keeping this in mind, rank the following compounds in order of descending acidity. PLEASE explain reasoning.arrow_forwardWhich compound would undergo carbocation rearrangement during an SN1 reaction? A B C Darrow_forward
- Rank the compounds in each group in order of increasing reactivity in electrophilic aromatic substitution: (a) C6H6, C6H5Cl, C6H5CHO, C6H5OCH3; (b) C6H5CH3, C6H5NH2, C6H5CH2NH2, C6H5CONH2.arrow_forwardConsider the tetracyclic aromatic compound drawn below, with rings labeled as A, B, C, and D. (a) Which of the four rings is most reactive in electrophilic aromatic substitution? (b) Which of the four rings is least reactive in electrophilic aromatic substitution? (c) What are the major product(s) formed when this compound is treated with one equivalent of Br2?arrow_forwardRank the following compounds in order of increasing reactivity in nucleophilic acyl substitution.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
