
ORGANIC CHEMISTRY-OWL V2 ACCESS
8th Edition
ISBN: 9781305582422
Author: Brown
Publisher: CENGAGE L
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 22, Problem 22.24P
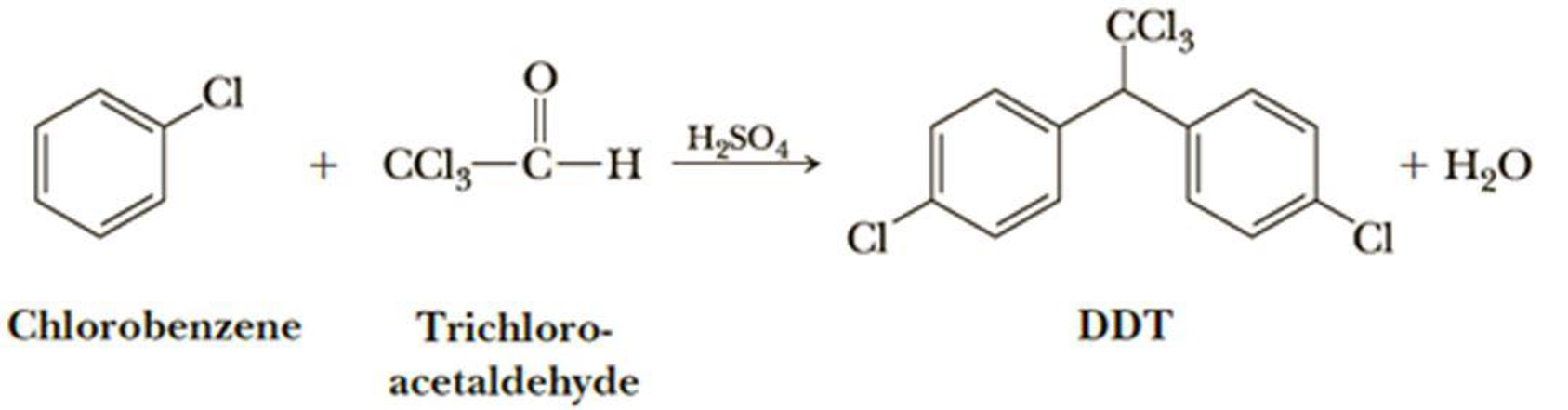
The insecticide DDT is prepared by the following route. Suggest a mechanism for this reaction. The abbreviation DDT is derived from the common name dichlorodiphenyltrichloroethane.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Suggest how you would synthesize each compound, use cyclopentanone as one of the reagents
Give the name and mechanism of this reaction asap
Propanal and propanone react in a similar way with acidified aqueous potassium cyanide, KCN. For this reaction to occur reasonably quickly, the pH of the solution should be approximately 4.
The reaction of propanal proceeds with acidified potassium cyanide proceeds more rapidly than that of propanone. Referring to the mechanism of the reactions, explain this phenomenon.
Chapter 22 Solutions
ORGANIC CHEMISTRY-OWL V2 ACCESS
Ch. 22.1 - Prob. 22.1PCh. 22.1 - Write a structural formula for the product from...Ch. 22.1 - Prob. 22.3PCh. 22.2 - Prob. 22.4PCh. 22.2 - Predict the major produce(s) of each electrophilic...Ch. 22.3 - In SN2 reactions of haloalkanes, the order of...Ch. 22 - Prob. 22.8PCh. 22 - Prob. 22.9PCh. 22 - Addition of m-xylene to the strongly acidic...Ch. 22 - Addition of tert-butylbenzene to the strongly...
Ch. 22 - What product do you predict from the reaction of...Ch. 22 - Other groups besides H+ can act as leaving groups...Ch. 22 - Prob. 22.14PCh. 22 - Prob. 22.15PCh. 22 - Prob. 22.16PCh. 22 - Prob. 22.17PCh. 22 - Suggest a reason why the nitroso group, N=O, is...Ch. 22 - Prob. 22.19PCh. 22 - Prob. 22.20PCh. 22 - The following molecules each contain two aromatic...Ch. 22 - Prob. 22.22PCh. 22 - Prob. 22.23PCh. 22 - The insecticide DDT is prepared by the following...Ch. 22 - Prob. 22.25PCh. 22 - Prob. 22.26PCh. 22 - Prob. 22.27PCh. 22 - Prob. 22.28PCh. 22 - Prob. 22.29PCh. 22 - Prob. 22.32PCh. 22 - Show how to prepare each compound from...Ch. 22 - Prob. 22.34PCh. 22 - Show reagents and conditions to bring about the...Ch. 22 - Prob. 22.36PCh. 22 - Propose a synthesis for each compound from...Ch. 22 - The first widely used herbicide for the control of...Ch. 22 - The first widely used herbicide for the control of...Ch. 22 - Prob. 22.40PCh. 22 - Prob. 22.41PCh. 22 - Prob. 22.42PCh. 22 - Prob. 22.43PCh. 22 - Cancer of the prostate is the second leading cause...Ch. 22 - Prob. 22.45PCh. 22 - Prob. 22.46PCh. 22 - Prob. 22.47PCh. 22 - When certain aromatic compounds are treated with...Ch. 22 - Prob. 22.49PCh. 22 - Following is the structure of miconazole, the...Ch. 22 - Prob. 22.51PCh. 22 - Prob. 22.52PCh. 22 - Prob. 22.53PCh. 22 - Show how the antidepressant venlafaxine (Effexor)...Ch. 22 - Prob. 22.57PCh. 22 - Given this retrosynthetic analysis, propose a...Ch. 22 - Prob. 22.59PCh. 22 - Prob. 22.60PCh. 22 - Prob. 22.61PCh. 22 - A newer generation of antipsychotics, among them...Ch. 22 - Prob. 22.63P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Enamines normally react with methyl iodide to give two products: one arising from alkylation at nitrogen and the second arising from alkylation at carbon. For example, Heating the mixture of C-alkylation and N-alkylation products gives only the product from C-alkylation. Propose a mechanism for this isomerization.arrow_forwardShow how to bring about each step in this synthesis of the herbicide propranil.arrow_forwardThe following molecule undergoes an intramolecular reaction in the presence of pyrrolidinium acetate, the protonated form of pyrrolidine. Draw the product of this reaction, assuming that a dehydration reaction takes place.arrow_forward
- Propanal and propanone react in a similar way with acidified aqueous potassium cyanide, KCN. For this reaction to occur reasonably quickly, the pH of the solution should be approximately 4. Draw a diagram to show the mechanism of the reaction of either propanal or propanone with acidified potassium cyanide.arrow_forwardThe reaction is a nucleophilic substitution, A is OH- and B is an alkyl bromide. Give the reaction mechanism and explain your reasoning.arrow_forwardGive step-by-step mechanism for the following reactionarrow_forward
- the following reactions will not produce the product given. briefly explain why.arrow_forwardPredict the major products formed when benzoyl chloride (PhCOCl) reacts with the following reagents.(a) ethanoarrow_forwardThe following substances can be prepared by a nucleophilic addition reaction between an aldehyde or ketoneand a nucleophile. Identify the reactants from which they were prepared. If the substance is an acetal, identifythe carbonyl compound and the alcohol; if it is an imine or enamine, identify the carbonyl compound and theamine. You do not have to consider stereochemistry. In cases where there is more than one answer, just giveone. Use Grignard reagents when an organometallic reagent is required. Draw the Grignard reagent as acovalent magnesium bromide.arrow_forward
- the organic compound 2-heptanone, belonging to the ketone family, is responsible for the strong penetrating odor in Roquefort cheeses. Starting from acetylene as the starting reagent, propose a synthesis line with the reaction mechanisms involved for the synthetic obtaining of 2-heptanone and use it as a food additive in analogous cheeses.arrow_forwardThe Friedel-Crafts Alkylation of p-xylene n-propyl bromide can result in an isopropyl as well as n-propyl substitution. A) Write the chemical equation involved in this reaction. B) Which will be the major and minor product? What do you expect will be the ratio of n-propyl to isopropyl substitution for p-xylene?arrow_forwardWhich reactions will produce the desired product in good yield? You may assume thataluminum chloride is added as a catalyst in each case. For the reactions that will not givea good yield of the desired product, predict the major products.Reagents Desired Product(a) benzene + n-butyl bromide n-butylbenzenearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License