
Concept explainers
Draw the product formed when pentanoic anhydride
a.
b.
(a)
Interpretation:
The product formed by the treatment of pentanoic anhydride
Concept introduction:
The replacement or substitution of one functional group with another functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons is known as a nucleophile. In nucleophilic acyl substitution reaction, a nucleophile takes the position of a leaving group.
Answer to Problem 22.46P
No product is formed by the treatment of pentanoic anhydride with
Explanation of Solution
Acid anhydride do not undergo reaction with
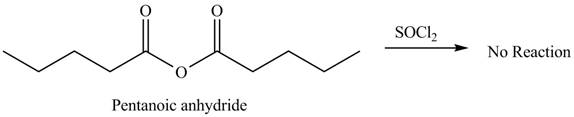
Figure 1
No product is formed by the treatment of pentanoic anhydride with
(b)
Interpretation:
The product formed by the treatment of pentanoic anhydride
Concept introduction:
The replacement or substitution of one functional group with another functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons is known as a nucleophile. In nucleophilic acyl substitution reaction, a nucleophile takes the position of a leaving group.
Answer to Problem 22.46P
The product formed by the treatment of pentanoic anhydride
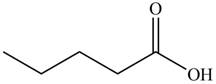
Explanation of Solution
Acid anhydride undergoes hydrolysis on treatment with
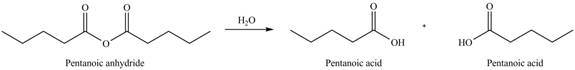
Figure 2
The product formed by the treatment of pentanoic anhydride
(c)
Interpretation:
The product formed by the treatment of pentanoic anhydride
Concept introduction:
The replacement or substitution of one functional group with another functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons is known as a nucleophile. In nucleophilic acyl substitution reaction, a nucleophile takes the position of a leaving group.
Answer to Problem 22.46P
The product formed by the treatment of pentanoic anhydride
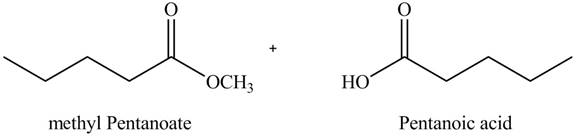
Explanation of Solution
Acid anhydride on reaction with alcohol gives one mole of ester and one mole of carboxylic acid. Methyl pentanoate and pentanoic acid are formed when pentanoic anhydride is treated with methanol as shown in Figure 3.
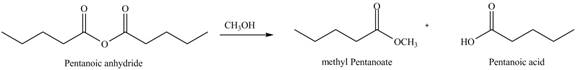
Figure 3
The product formed by the treatment of pentanoic anhydride
(d)
Interpretation:
The product formed by the treatment of pentanoic anhydride
Concept introduction:
The replacement or substitution of one functional group with another functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons is known as a nucleophile. In nucleophilic acyl substitution reaction, a nucleophile takes the position of a leaving group.
Answer to Problem 22.46P
No product is formed by the treatment of pentanoic anhydride with
Explanation of Solution
Acid anhydride do not undergo reaction with
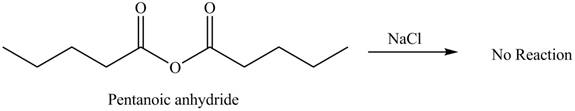
Figure 4
No product is formed by the treatment of pentanoic anhydride with
(e)
Interpretation:
The product formed by the treatment of pentanoic anhydride
Concept introduction:
The replacement or substitution of one functional group with another functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons is known as a nucleophile. In nucleophilic acyl substitution reaction, a nucleophile takes the position of a leaving group.
Answer to Problem 22.46P
The product formed by the treatment of pentanoic anhydride
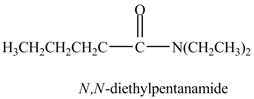
Explanation of Solution
The reaction of acid anhydride with secondary amine leads to the formation of amide. The nitrogen atom of
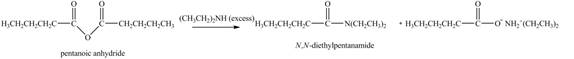
Figure 5
The product formed by the treatment of pentanoic anhydride
(f)
Interpretation:
The product formed by the treatment of pentanoic anhydride
Concept introduction:
The replacement or substitution of one functional group with another functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons is known as a nucleophile. In nucleophilic acyl substitution reaction, a nucleophile takes the position of a leaving group.
Answer to Problem 22.46P
The product formed by the treatment of pentanoic anhydride
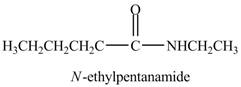
Explanation of Solution
The reaction of acid anhydride with primary amine leads to the formation of amide. The nitrogen atom of
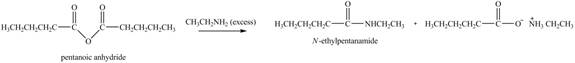
Figure 6
The product formed by the treatment of pentanoic anhydride
Want to see more full solutions like this?
Chapter 22 Solutions
ALEKS 360 CHEMISTRY ACCESS
- Draw the product formed when pentanal (CH3CH2CH2CH2CHO) is treatedwith following reagent. With some reagents, no reaction occurs. [1] HC≡CNa; [2] H2Oarrow_forwardDraw the product formed when (CH3)2CHOH is treated with following reagent. PBr3, then NaCNarrow_forwardDraw the product formed when (CH3)2CHOH is treated with following reagent. H2SO4arrow_forward
- Draw the product formed when pentanal (CH3CH2CH2CH2CHO) is treatedwith following reagent. With some reagents, no reaction occurs. [1] C6H5Li; [2] H2Oarrow_forwardDraw the product formed when (CH3)2CHOH is treated with following reagent. HBrarrow_forwardDraw the product formed when (CH3)2CHOH is treated with following reagent. TsCl, pyridinearrow_forward
- Draw the products formed when A or B is treated with each reagent. In some cases, no reaction occurs. [1] C6H5Li (excess); [2] H2Oarrow_forwardDraw the product formed when C6H5N2 +Cl− reacts with each compound.arrow_forwardDraw the product formed when C6H5N2+Cl− reacts with each compound.arrow_forward
- Draw the products formed when (CH3)2C=CH2 is treated with each reagent.a. HBrb. H2OH2SO4c. CH3CH2OH, H2SO4d. Cl2e. Br2, H2Of. NBS (aqueous DMSO)g. [1]BH3;[2]H2O2, HO-arrow_forwardDraw the products formed when D-altrose is treated with each reagent. a. (CH3)2CHOH, HCl b. NaBH4, CH3OH c. Br2, H2O d. HNO3, H2O e. [1] NH2OH; [2] (CH3CO)2O, NaOCOCH3; [3] NaOCH3 f. [1] NaCN, HCl; [2] H2, Pd-BaSO4; [3] H3O+ g. CH3I, Ag2O h. C6H5CH2NH2, mild H+arrow_forwardwhich reagents complete the reaction?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





