
Concept explainers
Interpretation:
The validation corresponding to the fact that data of carbon monoxide coverage and pressure follows a Langmuir isotherm is to be stated. The equilibrium constant for the adsorption process is to be stated.
Concept introduction:
The isotherm model that describes the adsorption process, if an adsorbate acts as an ideal gas at the isothermal situation is known as Langmuir adsorption isotherm model. In the given isothermal situation, the partial pressure of the adsorbate is related to the volume of the adsorbate which is adsorbed on the solid adsorbent.
Answer to Problem 22.47E
The occurrence of the linear straight line between
The equilibrium constant for the adsorption process is
Explanation of Solution
The given temperature is
The given data of carbon monoxide coverage and pressure in torr is mentioned in the table below.
| Coverage | Pressure (torr) |
The concentration of carbon monoxide is calculated by the formula given below.
Where,
•
•
•
•
•
•
The value of universal gas constant is
| Pressure | Coverage | |||
The graph between
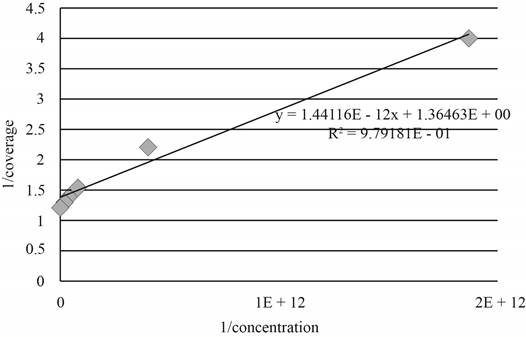
Figure 1
As, the plot between
The value of slope is predicted as
The value of equilibrium constant is calculated by the expression given below.
Substitute the value of slope in the above expression.
Therefore, the value of equilibrium constant is
The given data of carbon monoxide coverage and pressure follows a Langmuir isotherm.
The equilibrium constant for the adsorption process is
Want to see more full solutions like this?
Chapter 22 Solutions
Bundle: Physical Chemistry, 2nd + Student Solutions Manual
- Sodium hydride (NaH) crystallizes in the rock-salt structure, with four formula units of NaH per cubic unit cell. A beam of monoenergetic neutrons, selected to have a velocity of 2.639 × 103 ms-1 , is scattered in second order through an angle of 2u= 36.26° by the parallel faces ofthe unit cells of a sodium hydride crystal.(a) Calculate the wavelength of the neutrons.(b) Calculate the edge length of the cubic unit cell.(c) Calculate the distance from the center of an Na+ ion to the center of a neighboring H- ion.(d) If the radius of an Na+ ion is 0.98 Å, what is the radius of an H- ion, assuming the two ions are in contact?arrow_forwardOne of the chemical controversies of the nineteenth century concerned the element beryllium (Be). Berzelius originally claimed that beryllium was a trivalent element (forming Be3+ ions) and that it gave an oxide with the formula Be2O3. This resulted in a calculated atomic mass of 13.5 for beryllium. In formulating his periodic table, Mendeleev proposed that beryllium was divalent (forming Be2+ ions) and that it gave an oxide with the formula Be2O3. This assumption gives an atomic mass of 9.0. In 1894, A. Combes (Comptes Rendus 1894, p. 1221) reacted beryllium with the anion C5H7O2and measured the density of the gaseous product. Combess data for two different experiments are as follows: I II Mass 0.2022 g 0.2224 g Volume 22.6 cm3 26.0 cm3 Temperature 13C 17C Pressure 765.2 mm Hg 764.6 mm If beryllium is a divalent metal, the molecular formula of the product will be Be(C5H7O2)2; if it is trivalent, the formula will be Be(C5H7O2)3. Show how Combess data help to confirm that beryllium is a divalent metal.arrow_forwardGases produced by a chemical reaction can easily be collected over water. To determine the pressure of the dry gas, the vapor pressure of the water at that temperature must be subtracted from the total pressure. 1) Consider the following reaction: Mg(s) + 2 HC1(aq) → MgCl2(aq) + H2(g) The total pressure of gas collected over water is 725.0 mmHg and the temperature is 19.5°C What is the pressure of hydrogen gas formed in mmHg? Pressure = mmHgarrow_forward
- The Haber process is very important for agriculture because it converts N2(g) from the atmosphere into bound nitrogen, which can be taken up and used by plants. The Haber process reaction is N2(g) + 3 H2(g) = 2 NH3(g). The reaction is exo- thermic but is carried out at relatively high temperatures. Why?arrow_forwardCalculate the density of cesium if it crystallizes in the bcc structure with an atomic radius of 272 pm.arrow_forwardAt its critical point, ammonia has a density of 0.235 g cm-3 .You have a special thick-walled glass tube that has a10.0-mm outside diameter, a wall thickness of 4.20 mm, anda length of 155 mm. How much ammonia must you sealinto the tube if you wish to observe the disappearance of themeniscus as you heat the tube and its contents to a temperature higher than 132.23°C, the critical temperature?arrow_forward
- Polonium is the only element known to crystallize in the simple cubic lattice. (a) What is the distance between nearest neighbor polo- nium atoms if the first-order diffraction of X-rays with A = 1.785 Å from the parallel faces of its unit cells appears at an angle of 20 = 30.96° from these planes? (b) What is the density of polonium in this crystal (in g cm-3)?arrow_forwardGases produced by a chemical reaction can easily be collected over water. To determine the pressure of the dry gas, the vapor pressure of the water at that temperature must be subtracted from the total pressure. 1) Consider the following reaction: Mg(s) +2 HC1(aq) → MgCl2 (aq) + H2(g) The total pressure of gas collected over water is 725.0 mmHg and the temperature is 25.0°C What is the pressure of hydrogen gas formed in mmHg? Pressure = mmHgarrow_forwardProvide a particle level explanation for the following: when the temperature of a sealed, rigid (non-flexible) container is decreased, pressure also decreases.arrow_forward
- If the molecular weight of air is 28.9,what is the density of air at atmosphericpressure and a temperature of 368.7 K?1 atm = 1.013 × 10^5 N/m2, the mass ofa proton is 1.67262 × 10 ^−27 kg , Avogadro’snumber is 6.02214 × 10 ^23 mol−1and k =1.38065 × 10^ −23 N · m/K .Answer in units of kg/m3.arrow_forwardoxygen gas from the decomposition of KClO3 was collected by water displacement. the barometric pressure during the experiment was 723,9mmHg and the temperature was 20.0C. what is the partial pressure of oxygen collected (the vapor pressure of water at 20C is 17.5mmHg)?arrow_forwardPure gold crystallizes in a face-centered cubic unit cell with an edge length of 4.08 Å. The alloy called 18-karat gold consists of 75% Au, 8.0% Ag, and 17% Cu by mass. What are the subscripts in the empirical formula of 18-karat gold (Au(x)Ag(y)Cu(z)) to two significant digits?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





