
Concept explainers
Draw the structure corresponding to each name.
a.
b. isopropyl propanoate
c. acetic formic anhydride
d. N-isobutyl-N-methylbutanamide
e.
f. o-cyanobenzoic acid
g.
h. N-ethylhexanamide
(a)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: One should follow the given steps to draw the structure of a compound from its name. The first step is identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in the direction, where functional group gets a lower number. The third step is addition of substituents at appropriate atoms.
Answer to Problem 22.5P
The structure corresponding to the given name is,
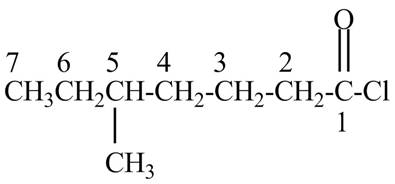
Explanation of Solution
The given name indicates that compound contains acid chloride (name ends with –yl chloride) as the functional group and heptane as the longest carbon chain with one methyl substitution at
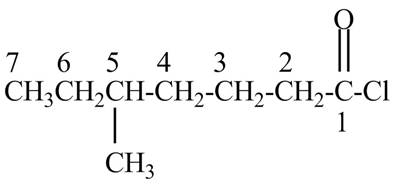
Figure 1
The structure corresponding to the given name is drawn in Figure 1.
(b)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: One should follow the given steps to draw the structure of a compound from its name. The first step is identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in the direction, where functional group gets a lower number. The third step is addition of substituents at appropriate atoms.
Answer to Problem 22.5P
The structure corresponding to the given name is,
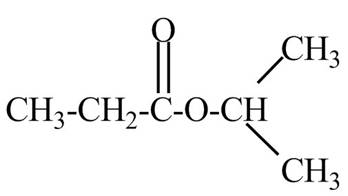
Explanation of Solution
The given name indicates that compound contains ester (name ends with –ate) as the functional group and propane as the longest carbon chain with one isopropyl substitution as
Thus, the structure of isopropyl propanoate is drawn as follows.
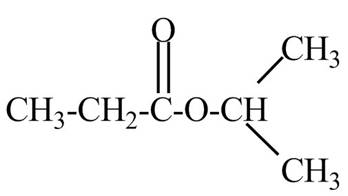
Figure 2
The structure corresponding to the given name is drawn in Figure 2.
(c)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: One should follow the given steps to draw the structure of a compound from its name. The first step is identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in the direction, where functional group gets a lower number. The third step is addition of substituents at appropriate atoms.
Answer to Problem 22.5P
The structure corresponding to the given name is,
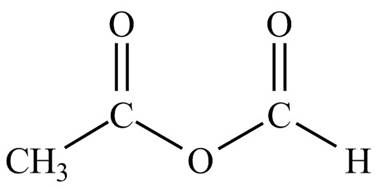
Explanation of Solution
The given name indicates that compound contains anhydride (name ends with -anhydride) as the functional group, which is derived from acetic acid and formic acid. Thus, it is a mixed anhydride. The structures of mixed anhydrides are drawn by joining the one ends of
Hence, the structure of acetic formic anhydride is drawn as follows.
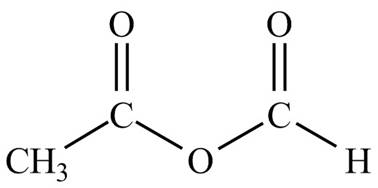
Figure 3
The structure corresponding to the given name is drawn in Figure 3.
(d)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: One should follow the given steps to draw the structure of a compound from its name. The first step is identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in the direction, where functional group gets a lower number. The third step is addition of substituents at appropriate atoms.
Answer to Problem 22.5P
The structure corresponding to the given name is,
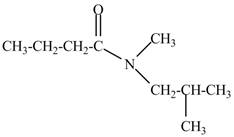
Explanation of Solution
The given name indicates that compound contains amide (name ends with -amide) as the functional group and butane as the longest carbon chain with one methyl and one isobutyl substitutions at
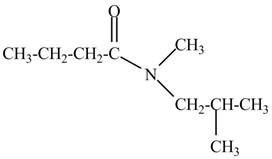
Figure 4
The structure corresponding to the given name is drawn in Figure 4.
(e)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: One should follow the given steps to draw the structure of a compound from its name. The first step is identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in the direction, where functional group gets a lower number. The third step is addition of substituents at appropriate atoms.
Answer to Problem 22.5P
The structure corresponding to the given name is,
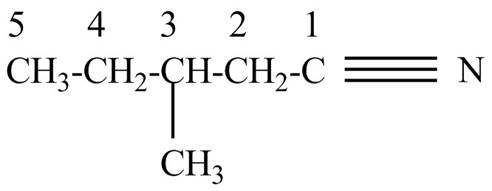
Explanation of Solution
The given name indicates that compound contains cyano (name ends with –nitrile) as the functional group and pentane as the longest carbon chain with one methyl substitution at

Figure 5
The structure corresponding to the given name is drawn in Figure 5.
(f)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: One should follow the given steps to draw the structure of a compound from its name. The first step is identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in the direction, where functional group gets a lower number. The third step is addition of substituents at appropriate atoms.
Answer to Problem 22.5P
The structure corresponding to the given name is,
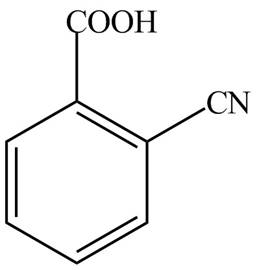
Explanation of Solution
The given name indicates that compound contains carboxylic acid (name ends with -oic acid) as the functional group, which is attached to the benzene ring with one cyano group at ortho position. Therefore, the structure of o-cyanobenzoic acid is drawn as follows.

Figure 6
The structure corresponding to the given name is drawn in Figure 6.
(g)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: One should follow the given steps to draw the structure of a compound from its name. The first step is identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in the direction, where functional group gets a lower number. The third step is addition of substituents at appropriate atoms.
Answer to Problem 22.5P
The structure corresponding to the given name is,
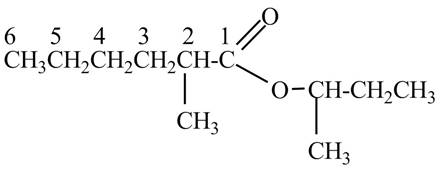
Explanation of Solution
The given name indicates that compound contains ester (name ends with –ate) as the functional group and hexane as the longest carbon chain with one methyl substitution at

Figure 7
The structure corresponding to the given name is drawn in Figure 7.
(h)
Interpretation: The structure corresponding to the given name is to be drawn.
Concept introduction: One should follow the given steps to draw the structure of a compound from its name. The first step is identification of parent name and functional group found at the end of the name. The second step is numbering of carbon skeleton in the direction, where functional group gets a lower number. The third step is addition of substituents at appropriate atoms.
Answer to Problem 22.5P
The structure corresponding to the given name is,
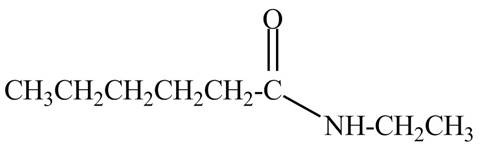
Explanation of Solution
The given name indicates that compound contains amide (name ends with -amide) as the functional group and hexane is the longest carbon chain with one ethyl substitution to
Therefore, the structure of N-ethylhexanamide is drawn as follows.

Figure 8
The structure corresponding to the given name is drawn in Figure 8.
Want to see more full solutions like this?
Chapter 22 Solutions
Study Guide/Solutions Manual for Organic Chemistry
- Amino acids such as glycine are the building blocks of large molecules called proteins that give structure to muscle, tendon, hair, and nails. a.) Explain why glycine does not actually exist in the form with all atomsuncharged, but actually exists as a salt called a zwitterion. b.) What product is formed when glycine is treated with concentratedHCl?c.) What product is formed when glycine is treated with NaOH?arrow_forward4. WHAT ARE IMINES? HOW ARE THEY FORMED? 5. WHAT IS GRIGNARD REACTION? WHAT CONSTITUTES A GRIGNARD REAGENT? 6. WHAT IS KETO-ENOL TAUTOMERIZATION?arrow_forwardGive the missing reagentsarrow_forward
- (plz with detaol explanation )arrow_forwardWhich of the following will not oxidize benzaldehyde? -- acidified KMnO4 -- acidified K2CrO4 -- Tollen’s reagent followed by H3O+ -- Basic I2 in KIarrow_forwardis ethyl acetate and benzoic acid in NaHCO3 soluble or insoluble? Please explain why lIs cyclohexene and etyhylamine soluble/insoluble in NaOH? Please explain why. Is Acetic anhydride soluble/ insoluble in HCl? Why? Is H2SO4 soluble/insoluble in Cyclohexene? Please explain whyarrow_forward
- As we have seen in this chapter, carbon-carbon double bonds are electron-rich regions that are attacked by electrophiles (e.g., HBr); they are not attacked by nucleophiles (e.g., diethylamine). However, when the carbon-carbon double bond has a carbonyl group adjacent to it, the double bond reacts readily with nucleophiles by nucleophilic addition (Section 19.8). Account for the fact that nucleophiles add to a carbon-carbon double bond adjacent to a carbonyl group and account for the regiochemistry of the reaction.arrow_forwardNot all aldehyde give a positve Bendicts test. Which of the follwing aldehydes do? a. d. b. e. c.arrow_forwardI need help with part G. Which was the formula of acetal product?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


