
Concept explainers
(1)
Interpretation:
The most acidic hydrogens in the given compounds are to be indicated.
Concept introduction:
The substances that produce hydrogen ions (
According to the definition of Bronsted and Lowry, acids are the substances that donate protons whereas bases are the substances that accept protons.
(1)
Answer to Problem 22.60SP
The most acidic hydrogens in the given compounds are shown in Figure 1.
Explanation of Solution
In a molecule, different types of protons are present. However, when acting as acids, only the most acidic proton participates in the acid-base reaction. The protons of functional groups are most acidic in nature as they are easily available for donation.
Acidic hydrogen is defined as that hydrogen which is attached to more electronegative atoms like oxygen and fluorine. The compound (f) does not contain any acidic hydrogen atom in the given compounds.
The most acidic hydrogens in the given compounds are shown below.
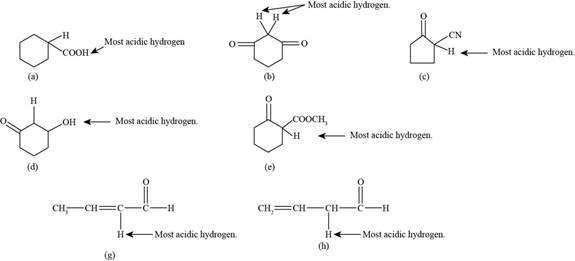
Figure 1
(2)
Interpretation:
The structures of important resonance contributors of the anions of the given compounds are to be drawn.
Concept introduction:
The delocalization of electrons due to presence of lone pair and double bond is called resonating structure. The structure is more stable if the resonating structures are more.
(2)
Answer to Problem 22.60SP
The structures of important resonance contributors of the anions of the given compounds are shown below.
Explanation of Solution
The formation of carbanion occurs when acidic hydrogen is removed from a compound. The important resonance contributors of the anions that results from the removal of most acidic hydrogen are shown below.
(a)
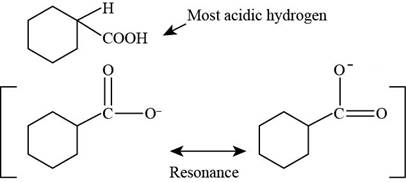
Figure 2
(b)
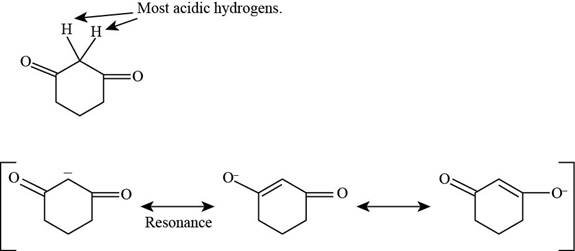
Figure 3
(c)
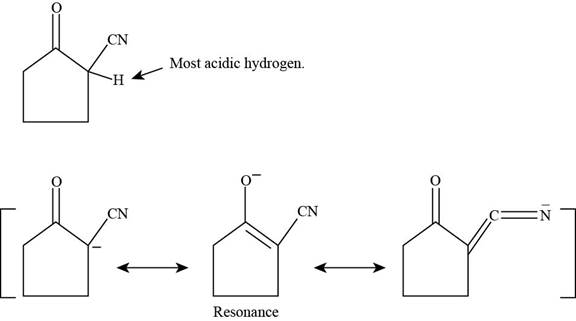
Figure 4
(d)
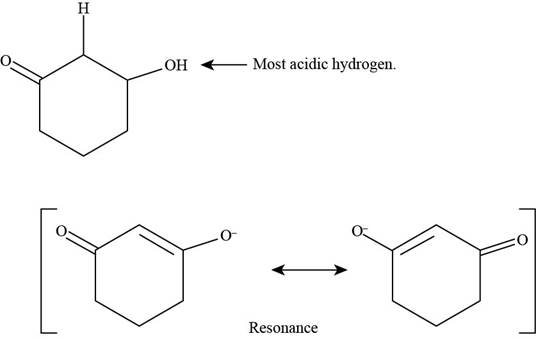
Figure 5
(e)
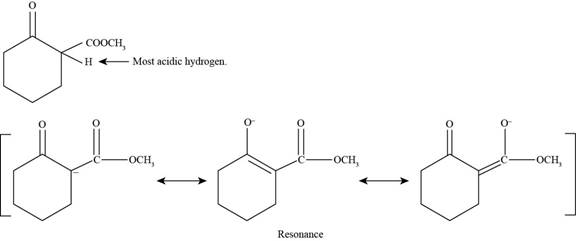
Figure 6
(f)
The given compound is shown below.
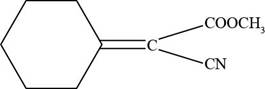
Figure 7
The given compound does not contain any acidic hydrogen atom. Therefore, it does not show resonance structures.
(g)
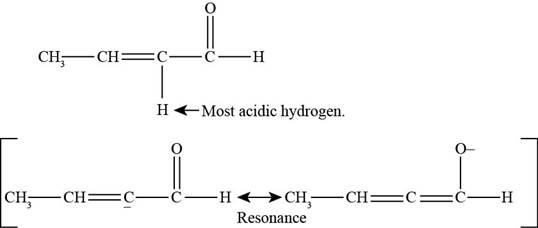
Figure 8
(h)
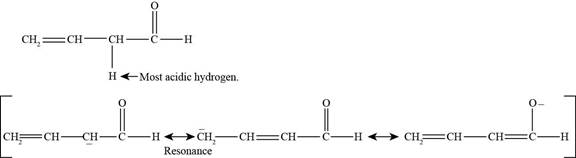
Figure 9
Want to see more full solutions like this?
Chapter 22 Solutions
ORGANIC CHEMISTRY 9E-W/MASTCHEMISTRY>CI
- Draw resonance contributors for each of the following species and rank them in order of decreasing contribution to the resonance hybrid. Then draw the resonance hybrid.arrow_forwarddraw the major organic product of the Bronsted acid-base reaction. Include all lone pairs and charges as appropriate. Ignore any counterions.arrow_forwardIn the picture, Each of the following compounds has more than one kind of (a-H's). First draw all a-H in, and circle the ones that are more acidic (that is, the ones more likely removed with a suitable base) in each compound. Thank you!arrow_forward
- For the following groups of molecules (labeled A-C), rank the acidity of the molecules in order from least acidic (3) to most acidic (1). Explain your reasoning.arrow_forwardAnswer is Reaction 7? Can you explain to me, please?arrow_forwardFor the following molecules: circle the most acidic hydrogen(s). determine the approximate pKa of the hydrogen you circled rank the compounds from least (1) to most (5) acidic compound.arrow_forward
- Answer each part for the reaction below Draw in ALL missing lone pairs Draw appropriate curved arrows to complete the acid-base reaction Draw the corresponding products for the reactionarrow_forwardHi! I need an explanation for this. More so, a technique. Technically, we're just separating the benzene from others? And this "others" would be the "acid chloride" needed to prepare for each ketone? Is that a trick that I can use for a long run? Please explain, thank you.arrow_forwardDraw a major resonance contributor of this enolate anion. Include all lone pairs in your structure.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
