
ORGANIC CHEMISTRY (LL)-W/STUDY GUIDE
8th Edition
ISBN: 9780134653631
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 22, Problem 32P
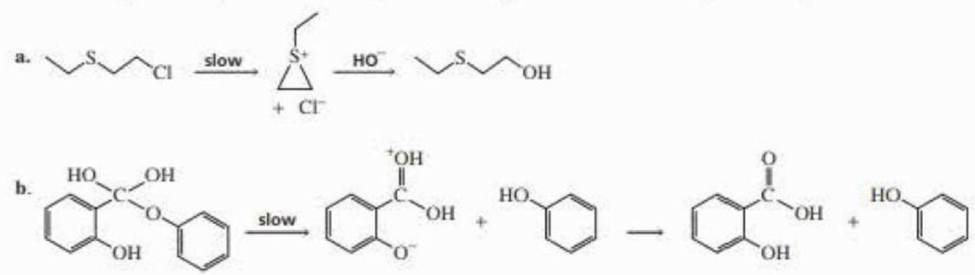
Indicate the type of catalysis that is occurring in the slow step in each of the following reaction sequences:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Indicate the type of catalysis that is occurring in the slow step in each of the following reaction sequences:
In each of the following three reaction coordinate diagrams, state:
Q) Whether the reaction is the slowest, the fastest, or intermediate in rate
Which of the following reactions occurs by a one-step mechanism as opposed to a two-step mechanism?
Chapter 22 Solutions
ORGANIC CHEMISTRY (LL)-W/STUDY GUIDE
Ch. 22.2 - Compare each of the mechanisms listed here with...Ch. 22.2 - Prob. 3PCh. 22.2 - Prob. 4PCh. 22.3 - a. Draw the mechanism for the following reaction...Ch. 22.5 - Prob. 7PCh. 22.5 - Propose a mechanism for the Co2+ catalyzed...Ch. 22.6 - Prob. 9PCh. 22.7 - Prob. 10PCh. 22.7 - Prob. 12PCh. 22.7 - Prob. 13P
Ch. 22.9 - Which of the following amino acid side chains can...Ch. 22.9 - Which of the following C-terminal peptide bonds is...Ch. 22.9 - Carboxypeptidase A has esterase activity as well...Ch. 22.10 - Arginine and lysine side chains fit into trypsins...Ch. 22.10 - Explain why serine proteases do not catalyze...Ch. 22.11 - If H2 18O is used in the hydrolysis reaction...Ch. 22.11 - Draw the pH-activity profile for an enzyme that...Ch. 22.12 - The pHactivity profile for glucose-6-phosphate...Ch. 22.12 - Prob. 23PCh. 22.13 - Draw the mechanism for the hydroxide ion-catalyzed...Ch. 22.13 - What advantage does the enzyme gain by forming an...Ch. 22.13 - Prob. 26PCh. 22.13 - Prob. 27PCh. 22.13 - Aldolase shows no activity if it is incubated with...Ch. 22 - Which of the following parameters would be...Ch. 22 - Prob. 29PCh. 22 - Prob. 30PCh. 22 - Prob. 31PCh. 22 - Indicate the type of catalysis that is occurring...Ch. 22 - The deuterium kinetic isotope effect (KH2O/KD2O)...Ch. 22 - Prob. 34PCh. 22 - Co2+ catalyzes the hydrolysis of the lactam shown...Ch. 22 - there are two kinds of aldolases. Class I...Ch. 22 - Prob. 37PCh. 22 - The hydrolysis of the ester shown here is...Ch. 22 - Prob. 39PCh. 22 - At pH = 12, the rate of hydrolysis of ester A is...Ch. 22 - 2-Acetoxycyclohexyl tosylate reacts with acetate...Ch. 22 - Proof that an imine was formed between aldolase...Ch. 22 - Prob. 43PCh. 22 - a. Explain why the alkyl halide shown here reacts...Ch. 22 - Triosephosphate isomerase (TIM) catalyzes the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In each of the following, which reaction mechanism assumption is apparently being violated? Explain your answers. a. A reaction takes place more rapidly when the concentration of reactants is decreased. b. A reaction takes place more rapidly when the reaction mixture is cooled. c. The reaction rate of A+BAB increases as the concentration of A is increased but does not change as the concentration of B is increased.arrow_forwardWhich of the four reactions does NOT proceed through the same intermediate as the others?arrow_forwardWrite a rate equation for each reaction, given the indicated mechanism.arrow_forward
- What is the type of catalysis is in the given reaction: CH3 – CH = CH2(g) + H2(g) Ni (s) CH3CH2CH3(g). Homogenous Catalysis Haber Process Contact Process Catalysis Hydrogenation Catalysisarrow_forwardWhat is the mechanism, step by step, for this reaction?arrow_forwardCarbonic Anhydrase or carbonic dehydratase is a family of metalloenzymes containingzinc (Zn2+) ion in its active site. Carbonic anhydrase can greatly increase the rate of thisreaction, reaching a reaction rate of 104 - 106 per second. It is defined as the enzyme found inred blood cells, other parts of animals and plants, that breaks down carbonic acid with carbondioxide and water.CO2(g) + H2O(l) → H2CO3(aq)For the carbonic anhydrase activity specified by the equation above, evaluate the standardreaction Gibbs energy at 25 °C?arrow_forward
- Explain the Energy Diagram for a Two-Step Reaction Mechanism ?arrow_forwardDraw the reaction coordinate of catalyzed and uncatalyzed reactions. Indicate the substrate (S), product (P), activation energy (∆G+), transition state (T), and free energy change (∆G°).arrow_forwardThe first graph below represents the energy level diagram for an uncatalyzed reaction. Which other graph could represent the same reaction but catalyzed?arrow_forward
- Chemistry Draw the structure of the missing reactants intermediate or products in the following mechanism. will rate.arrow_forwardAn increase in temperature will Group of answer choices decrease the rate of a reaction. decrease the activation energy for a reaction. increase the amount of collisions in the system. increase the number of elementary steps in the reaction. none of the above.arrow_forwardWhat is the instantaneous rate of the reaction?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License