
Chemistry: Principles and Practice
3rd Edition
ISBN: 9780534420123
Author: Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 22, Problem 45QE
(a)
Interpretation Introduction
Interpretation:
The given compound has to be identified based on the class of organic compounds.
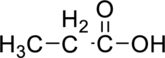
Concept Introduction:
Organic compounds are frequently categorized by certain groups of atoms that have characteristics chemical properties. A functional group is an atom or small group of atoms in a molecule that undergoes characteristic reaction.
(b)
Interpretation Introduction
Interpretation:
The given compound has to be identified based on the class of organic compounds.
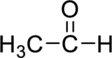
Concept Introduction:
Refer to sub part-a.
(c)
Interpretation Introduction
Interpretation:
The given compound has to be identified based on the class of organic compounds.
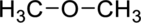
Concept Introduction:
Refer to sub part-a.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 22 Solutions
Chemistry: Principles and Practice
Ch. 22 - Prob. 1QECh. 22 -
Explain what is special about the element carbon...Ch. 22 - Prob. 3QECh. 22 - Prob. 4QECh. 22 - Prob. 5QECh. 22 - Prob. 6QECh. 22 - Prob. 7QECh. 22 - Prob. 8QECh. 22 - Prob. 9QECh. 22 - Prob. 10QE
Ch. 22 - Prob. 11QECh. 22 - Prob. 12QECh. 22 - Prob. 13QECh. 22 - Prob. 14QECh. 22 - Prob. 15QECh. 22 - Prob. 16QECh. 22 - Prob. 17QECh. 22 - Prob. 18QECh. 22 - Prob. 19QECh. 22 - Prob. 20QECh. 22 - Prob. 21QECh. 22 - Prob. 22QECh. 22 - Prob. 23QECh. 22 - Prob. 24QECh. 22 - Prob. 25QECh. 22 - Prob. 26QECh. 22 - Prob. 27QECh. 22 - Prob. 28QECh. 22 - Prob. 29QECh. 22 - Prob. 30QECh. 22 - Prob. 31QECh. 22 - Prob. 32QECh. 22 - Prob. 33QECh. 22 - Prob. 34QECh. 22 - Prob. 35QECh. 22 - Prob. 36QECh. 22 - Prob. 37QECh. 22 - Prob. 38QECh. 22 - Prob. 39QECh. 22 - Prob. 40QECh. 22 - Prob. 41QECh. 22 - Prob. 42QECh. 22 - Prob. 43QECh. 22 - Prob. 44QECh. 22 - Prob. 45QECh. 22 - Prob. 46QECh. 22 - Prob. 47QECh. 22 - Prob. 48QECh. 22 - Prob. 49QECh. 22 - Prob. 50QECh. 22 - Prob. 51QECh. 22 - Prob. 52QECh. 22 - Prob. 53QECh. 22 - Prob. 54QECh. 22 - Name the following compounds. (a) FCH2CH2CH2OH (b)...Ch. 22 - Prob. 56QECh. 22 - Prob. 57QECh. 22 - Prob. 58QECh. 22 - Prob. 59QECh. 22 - Prob. 60QECh. 22 - Prob. 61QECh. 22 - Prob. 62QECh. 22 - Prob. 63QECh. 22 - Prob. 64QECh. 22 - Prob. 65QECh. 22 - Prob. 66QECh. 22 - Prob. 67QECh. 22 - Prob. 68QECh. 22 - Prob. 69QECh. 22 - Prob. 70QECh. 22 - Prob. 71QECh. 22 - Prob. 72QECh. 22 - Prob. 73QECh. 22 - Prob. 74QECh. 22 - Prob. 75QECh. 22 - Prob. 76QECh. 22 - Prob. 77QECh. 22 - Prob. 78QECh. 22 - Prob. 79QECh. 22 - Prob. 80QECh. 22 - Prob. 81QECh. 22 - Prob. 82QECh. 22 - Prob. 83QECh. 22 - Prob. 84QECh. 22 - Prob. 85QECh. 22 - Prob. 86QECh. 22 - Prob. 87QECh. 22 - Prob. 88QE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- . Alkenes and alkynes are characterized by their ability to undergo rapid, complete reactions, by which other atoms attach themselves to the carbon atoms of the double or triple bond.arrow_forwardWrite structures for the three isomers of the aromatic hydrocarbon xylene, C6H4(CH3)2.arrow_forwardSelect those compounds that can be correctly called unsaturated and classify each one as an alkene or an alkyne: a.CH3CH2CH3f. b.CH3CH=CHCH3g. c.h.CH2=CHCH2CH3 d.i. e.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning



Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

World of Chemistry
Chemistry
ISBN:9780618562763
Author:Steven S. Zumdahl
Publisher:Houghton Mifflin College Div

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning