
ORGANIC CHEMISTRY
6th Edition
ISBN: 9781260826791
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 22.8, Problem 25P
What starting materials are needed to prepare each compound by the Michael reaction?
a.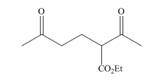 b.
b. 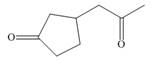
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What are the composition of the following reagents?
a. Tollen's reagent
b. Fehling's A
c. Fehling's B
d. Schiff's reagent
e. KMnO4
1. Which ones of the following compounds can be enolized?
4.
2
A. Compound 1 only
B. Compound 2 only
C. Compounds 1 and 2
D. Compounds 2 and 3
E. Compounds 2, 3, and 4
Br
Draw the products of each of the following reactions:
a.
b.
j
Grubbs
catalyst
Grubbs
catalyst
Grubbs
catalyst
Chapter 22 Solutions
ORGANIC CHEMISTRY
Ch. 22.1 - Prob. 1PCh. 22.1 - Prob. 2PCh. 22.1 - Problem 24.3
What unsaturated carbonyl compound is...Ch. 22.1 - Prob. 4PCh. 22.1 - Prob. 5PCh. 22.2 - Prob. 6PCh. 22.2 - Problem 24.7
Draw the products formed in each...Ch. 22.4 - Prob. 15PCh. 22.4 - Prob. 16PCh. 22.5 - Problem 24.16
What ester is formed when each...
Ch. 22.6 - Prob. 18PCh. 22.6 - Prob. 19PCh. 22.6 -
Draw the products of each reaction.
a. b.
Ch. 22.6 - Problem 24.20
Two steps in a synthesis of the...Ch. 22.7 - Prob. 22PCh. 22.8 - Problem 24.22
Which of the following compounds can...Ch. 22.8 - Prob. 24PCh. 22.8 - Problem 24.24
What starting materials are needed...Ch. 22 - Prob. 29PCh. 22 - 24.29 What steps are needed to convert A to B?
Ch. 22 - Prob. 31PCh. 22 - 24.31 Draw the product formed in each directed...Ch. 22 - Prob. 33PCh. 22 - 24.33 What starting materials are needed to...Ch. 22 - Prob. 35PCh. 22 - Prob. 36PCh. 22 - 24.36 Identify the structures of C and D in the...Ch. 22 - Prob. 38PCh. 22 - Prob. 39PCh. 22 - 24.39 Draw the product formed from a Claisen...Ch. 22 - Prob. 41PCh. 22 - 24.41 Even though B contains three ester groups, a...Ch. 22 - Prob. 43PCh. 22 - Prob. 44PCh. 22 - 24.44 Vetivone is isolated from vetiver, a...Ch. 22 - Draw the product of each Robinson annulation from...Ch. 22 - Prob. 50PCh. 22 - Prob. 51PCh. 22 - 24.52 Draw a stepwise mechanism for the following...Ch. 22 - Prob. 53PCh. 22 - Prob. 54PCh. 22 - Prob. 55PCh. 22 - Prob. 56PCh. 22 - Prob. 57PCh. 22 - Prob. 58PCh. 22 - Prob. 59PCh. 22 - 24.62 Devise a synthesis of each compound from ,...Ch. 22 - Prob. 63P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the major organic product obtained from the following reaction? A. B. HN il ΗΝ 'N' N C. D. Narrow_forward16.41 Draw the products of each reaction. a. b. HO NO₂ mellanrios H HNO3 H₂SO4 Br₂ FeBr3 AICI3 d. e. Br- edmun adtonie F prier f. F (X) napoln NO₂ Cl₂ FeCl3 SO3 H₂SO4 onho sur + Na Sarrow_forwardB. Give the IUPAC name for each compound. a. b. d.arrow_forward
- Compound that is most easily hydrolyzed by acid in water `NH A. В. F C. D.arrow_forwardWhat is the major organic product obtained from the following reaction? A. B. HN 1. H₂NCH3, H* cat. 2. NaBH3CN C. D. HNarrow_forwardElectron inductive effect of alkyl groups decreases the acidity in: A. acetic acid B. benzoic acid C. hydrochloric acid D nitric acidarrow_forward
- Several diamines are building blocks for the synthesis of pharmaceuticals and agro-chemicals. Show how both 1,3-propanediamine and 1,4-butanediamine can be prepared from acrylonitrile.arrow_forward2. How many different ß-hydroxyaldehydes and ß-hydroxyketones, including constitutional isomers and stereoisomers, are formed upon treatment of a mixture of acetone and benzaldehyde with base? a. b. 2 c. 3 d. 4arrow_forward17, The conversion of cyclohexene to cyclohexane requires: A. HCI B. NaOH C. H₂O and an acid catalyst D. H, and a catalystarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY