
ORGANIC CHEMISTRY-EBOOK>I<
9th Edition
ISBN: 9781305084414
Author: McMurry
Publisher: INTER CENG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 22.SE, Problem 30MP
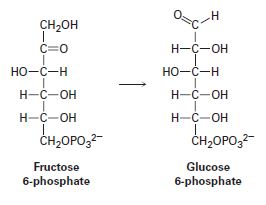
One of the later steps in glucose biosynthesis is the isomerization of fructose 6-phosphate to glucose 6-phosphate. Propose a mechanism, using acid or base catalysis as needed.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
One step in the gluconeogenesis pathway for the biosynthesis of glucose is the partial reduction of 3-phosphoglycerate to give glyceraldehyde 3-phosphate. The process occurs by phosphorylation with ATP to give 1,3-bisphosphoglycerate,
reaction with a thiol group on the enzyme to give an enzyme-bound thioester, and reduction with NADH.
-OPO3²- Enz-SH
H-C-OH
ATP
CH₂OPO3²-
3-phosphoglycerate
O
0-0--0
O
ADP
CH₂CH3
substitute for
1,3-bisphosphoglycerate
C
H-C-OH
CH₂OPO3²-
1,3-bisphosphoglycerate
O=C
CH3-SH
substitute for
Enz-SH
H
H-C-OH
|
CH₂OPO3²-
PO4³-
O.
S-Enz
H-C-OH
glyceraldehyde 3-phosphate
Propose a structure for the first intermediates in the reaction of 1,3-bisphosphoglycerate with a thiol group on the enzyme to form an enzyme-bound thioester. Assume a basic group on the enzyme catalyzes the formation of this
intermediate.
To simplify the drawing process, substitute the structures below for the 1,3-bisphosphoglycerate and Enz-SH.
CH₂OPO3²-
(Enzyme-bound
thioester)
NADH/H*
NAD*,…
The rate constant for the uncatalyzed reaction of two molecules of glycine ethyl ester to form glycylglycine ethyl ester is 0.6 M-1s-1. In the presence of Co2+, the rate constant is 1.5 x 106 M-1s-1. What rate enhancement does the catalyst provide?
In the biosynthesis of aromatic amino acids, erythrose-4-phosphate undergoes electrophilic addition to
phosphoenolpyruvate (PEP). Draw the products of this step, paying particular attention to regiochemistry.
ОН
OPOH
ОН
?
Enzyme
Erythrose 4-phosphate
Phosphoenolpyruvate
(PEP)
Chapter 22 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
Ch. 22.1 - Prob. 1PCh. 22.1 - How many acidic hydrogens does each of the...Ch. 22.1 - Prob. 3PCh. 22.3 - Write the complete mechanism for the deuteration...Ch. 22.3 - Prob. 5PCh. 22.4 - If methanol rather than water is added at the end...Ch. 22.5 - Prob. 7PCh. 22.5 - Draw a resonance structure of the acetonitrile...Ch. 22.6 - If methanol rather than water is added at the end...Ch. 22.7 - Prob. 10P
Ch. 22.7 - Draw a resonance structure of the acetonitrile...Ch. 22.7 - Why do you suppose ketone halogenations in acidic...Ch. 22.7 - Prob. 13PCh. 22.7 - Prob. 14PCh. 22.7 - Prob. 15PCh. 22.7 - Prob. 16PCh. 22.SE - Prob. 17VCCh. 22.SE - Prob. 18VCCh. 22.SE - Prob. 19VCCh. 22.SE - Prob. 20MPCh. 22.SE - Predict the product(s) and provide the mechanism...Ch. 22.SE - Predict the product(s) and provide the mechanism...Ch. 22.SE - Prob. 23MPCh. 22.SE - In the Hell–Volhard–Zelinskii reaction, only a...Ch. 22.SE - Prob. 25MPCh. 22.SE - Nonconjugated , -unsaturated ketones, such as...Ch. 22.SE - Prob. 27MPCh. 22.SE - Using curved arrows, propose a mechanism for the...Ch. 22.SE - Prob. 29MPCh. 22.SE - One of the later steps in glucose biosynthesis is...Ch. 22.SE - The Favorskii reaction involves treatment of an...Ch. 22.SE - Treatment of a cyclic ketone with diazomethane is...Ch. 22.SE - Prob. 33MPCh. 22.SE - Amino acids can be prepared by reaction of alkyl...Ch. 22.SE - Amino acids can also be prepared by a two-step...Ch. 22.SE - Heating carvone with aqueous sulfuric acid...Ch. 22.SE - Identify all the acidic hydrogens (pKa 25) in the...Ch. 22.SE - Rank the following compounds in order of...Ch. 22.SE - Prob. 39APCh. 22.SE - Base treatment of the following , -unsaturated...Ch. 22.SE - Prob. 41APCh. 22.SE - Prob. 42APCh. 22.SE - Prob. 43APCh. 22.SE - Which, if any, of the following compounds can be...Ch. 22.SE - Prob. 45APCh. 22.SE - Prob. 46APCh. 22.SE - Prob. 47APCh. 22.SE - How might you convert geraniol into either ethyl...Ch. 22.SE - Prob. 49APCh. 22.SE - One way to determine the number of acidic...Ch. 22.SE - Prob. 51APCh. 22.SE - Prob. 52APCh. 22.SE - Prob. 53APCh. 22.SE - Prob. 54APCh. 22.SE - Prob. 55APCh. 22.SE - Prob. 56APCh. 22.SE - All attempts to isolate primary and secondary...Ch. 22.SE - How would you synthesize the following compounds...Ch. 22.SE - Prob. 59APCh. 22.SE - Prob. 60APCh. 22.SE - Prob. 61APCh. 22.SE - Prob. 62APCh. 22.SE - As far back as the 16th century, South American...Ch. 22.SE - The key step in a reported laboratory synthesis of...Ch. 22.SE - Prob. 65AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- One of the steps in the pentose phosphate pathway for glucose catabolism is the reaction of sedoheptulose 7-phosphate with glyceraldehydes 3-phosphate in the presence of a transaldolase to yield erythrose 4-phosphate and fructose 6-phosphate. (a) The first part of the reaction is the formation of a protonated Schiff base of sedoheptulose 7-phosphate with a lysine residue in the enzyme followed by a retro-aldol cleavage to give an enamine plus erythrose 4-phosphate. Show the structure of the enamine and the mechanism by which it is formed. (b) The second part of the reaction is a nucleophilic addition of the enamine to glyceraldehyde 3-phosphate followed by hydrolysis of the Schiff base to give fructose 6-phosphate. Show the mechanism.arrow_forwardrivatives: Nucleophilic Acyl Substitution Reactions - EOC O C-O [References] One step in the gluconeogenesis pathway for the biosynthesis of glucose is the partial reduction of 3-phosphoglycerate to give glyceraldehyde 3-phosphate. The process occurs by phosphorylation with ATP to give 1,3-bisphosphoglycerate, reaction with a thiol group on the enzyme to give an enzyme-bound thioester, and reduction with NADH. H-C-OH ATP ADP CH₂OPO32- 3-phosphoglycerate 0= OPO32- C H-C-OH CH₂OPO32 1,3-bisphosphoglycerate Enz-SH PO43- O= C S-Enz H-C-OH CH₂OPO3² (Enzyme-bound thioester) NADH/H+ O=C NAD*, Enz-SH H H-C-OH CH₂OPO3²- glyceraldehyde 3-phosphate Propose a structure for the first intermediate in the reaction of the enzyme-bound thioester with NADH to form glyceraldehyde 3-phosphate. To simplify the drawing process, substitute the structure below for the enzyme-bound thioester. S-CH3 substitute for the CH2CH3 enzyme-bound thioester You do not have to consider stereochemistry. • You do not have…arrow_forwardAll the glucose units in dextran have six-membered rings. When a sample of dextran is treated with methyl iodide and Ag2O and the product ishydrolyzed under acidic conditions, the final products are 2,3,4,6-tetra-O-methyl-d-glucose, 2,4,6-tri-O-methyl-d-glucose, 2,3,4-tri-O-methyl-d-glucose, and 2,4-di-O-methyl-d-glucose. Draw a short segment of dextran.arrow_forward
- Ribose, a carbohydrate with the formula shown, forms a cyclic hemiacetal, which, in principle, could contain either a four- membered, five-membered, or six-membered ring. To determine which ring is formed, ribose is treated with methanol in the presence of an acid catalyst. The products are then isolated and treated with NaIO4 then with H3O+. OH HO OH OH Ribose, C5H10O5 H MeOH H* A & B isomeric cyclic acetals with formula C6H₁2O5 1. NalO4 2. H₂O* MeOH products Assuming that ribose formed a six-membered ring cyclic hemiacetal, draw the structure of the sodium periodate digestion products. • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner.arrow_forwardGive the major organic product that is formed when the primary hydroxyl group of the following monosaccharide undergo nucleophilic substitution reaction with acetic anhydridearrow_forwardA graduate student was studying enzymatic reductions of cyclohexanones when she encountered some interesting chemistry. When she used an enzyme and NADPH to reduce the following ketone, she was surprised to find that the product was optically active. She carefully repurified the product so that no enzyme, NADPH, or other contaminants were present. Still, the product was optically active. Is the product capable of showing optical activity? If it is, explain how.arrow_forward
- (a) What is the difference between the hormones progesterone and testosterone? (b) Draw the structure of a a steroid nucleus. (c) Give the products obtained from complete base hydrolysis in the following reaction: O || CH,−O−C−(CH2)14–CH3 O CH–0–C−(CH2)14—CH3 + 3 NaOH O CH,−0–C−(CH2)14–CH3arrow_forwardDraw the product formed when CH3CH2C=CH is treated with each of the following sets of reagents: (a) H2O, H2SO4, HgSO4; and (b) R2BH, followed by H2O2, HO−.arrow_forwardA chemist synthesized compound X as a racemic mixture. When the ketone group in X was enzymatically reduced to the corresponding alcohol, a 100% yield was obtained of the product shown below. Choose the statement that best describes this result. ОН enzyme C;H1 `OCH,CH; pH 4.0 C3H1 `OCH,CH3 ОН ÕH X (racemic) (100% yield) One enantiomer of compound X reacts quickly with the enzyme. The other enantiomer of compound X is unreactive, but rapidly equilibrates with the reactive enantiomer under the reaction conditions. Since compound X was racemic, it makes sense that only a single product was obtained. O The product is a meso compound, so either enantiomer of compound X gives the same product. One enantiomer of compound X reacts quickly with the enzyme, while the other enantiomer of compound X remains unchanged.arrow_forward
- A graduate student was studying enzymatic reductions of cyclohexanones when she encountered some interesting chemistry. When she used an enzyme and NADPH to reduce the following ketone, she was surprised to find that the product was optically active. She carefully repurified the product so that no enzyme, NADPH, or other contaminants were present. Still, the product was optically active. If this reaction could be accomplished using H2 and a nickel catalyst, would the product be optically active? Explain.arrow_forwardGive the name (common name or IUPAC name) of intermediate product (a) in the following reaction.arrow_forward2-Butanone is reduced by hydride ion donors, such assodium borohydride (NaBH₄), to the alcohol 2-butanol. Eventhough the alcohol has a chiral center, the product isolated from the redox reaction is not optically active. Explain.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY