
Concept explainers
How does the metal ion in carboxypeptidase A increase the enzyme’s catalytic activity
Interpretation:
The metal ion in carboxypetidase A increases the catalytic activity of the enzyme; this has to be explained.
Explanation of Solution
Carboxypetidase A is a digestive enzyme. It hydrolyzes the carboxyl terminal peptide bond and it prefers bulky and aliphatic residues.
Proposed mechanism for the catalytic reaction of carboxypetidase is given in figure 1.
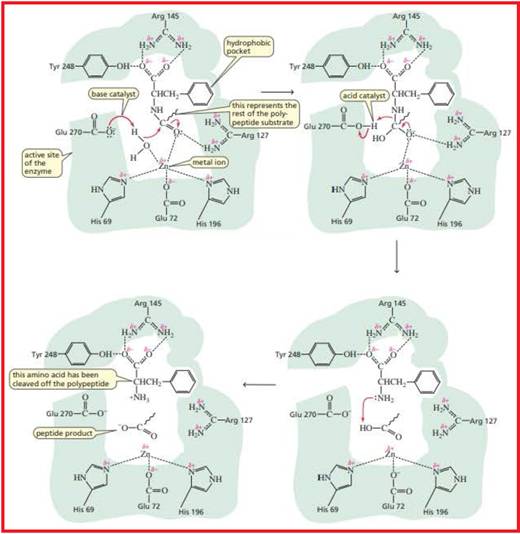
Figure 1
The metal ion present in the carboxypeptidase A enzyme is Zinc metal.
This metal ion
Want to see more full solutions like this?
Chapter 23 Solutions
ORGANIC CHEMISTRY-W/S.G+SOLN.MANUAL
- Describe the difference between graphs showing temperature versus reaction rate for an enzyme-catalyzed reaction and an uncatalyzed reaction.arrow_forwardThe site on an enzyme molecule that does the catalytic work is called the: a. binding site. b. allosteric site. c. lock. d. active site.arrow_forwardThe process by which an enzyme acts on the substrate can be described by the: a. lock-and-key model. b. enzyme-and-substrate model. c. enzyme folding model. d. catalytic model.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





