
Concept explainers
(a)
Interpretation:
The possibility of cis-trans isomer of Prop-1-ene is to be determined and if it exists, it needs to be drawn.
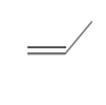
Concept introduction:
The cis-trans isomers, also known as geometric isomers, are isomers which have same molecular formula but have different permanent position in space.
(b)
Interpretation:
The possibility of cis-trans isomer of 2-Bromo-3-chlorobut-2-ene is to be determined and if it exists then it needs to be drawn.
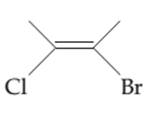
Concept introduction:
The cis-trans isomers, also known as geometric isomers, are isomers which have same molecular formula but have different permanent position in space.
(c)
Interpretation:
The possibility of cis-trans isomer of 1-Bromo-1-chloro-2-methylpropene is to be determined and if it exists then it needs to be drawn.
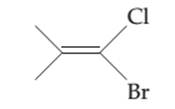
Concept introduction:
The cis-trans isomers, also known as geometric isomers, are isomers which have same molecular formula but have different permanent position in space.
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
CHEMISTRY-MASTERINGCHEMISTRY W/ETEXT
- Isomers are different compounds that have the same molecular formula. If the atoms are connected in different ways, they are called constitutional or structural isomers. Geometric isomers are a type of isomer where the order of the atoms in the two compounds is the same but their arrangement in space is different. The most common types of geometric isomers are cis- and trans- isomers. PART 5: Constitutional/Structural Isomers Draw (using any method you wish) and give the IUPAC name for any four isomers with the molecular formula C6H12Brz. Structure: - Br CH₂ сна CH₂CH CH CH₂ CH₂ Br. Name: bromo-3 methyl peature Name: Structure: Structure: Name: Structure: comors Name:arrow_forwardWhich is most stable, cis-1,4-dimethylhexane, or the trans isomoer? Draw the structure of the more stable isomer (your structure should clearly distinguish between all axial and equatorial positions.)arrow_forwardDraw the skeletal ("line") structure of an isomer of this molecule: HO OHarrow_forward
- Write the letter T or F inside each box after deciding which statement is true or false. Then on the 3rd box, write the letter of your choice based on the given instructions. A if both statements are true. B if the 1st statement is false and the 2nd statement is true. C if the 1st statement is true and the 2nd statement is false. D if both statements are falsearrow_forwardlist at least three tests that can be utilized to test the health of the cardiovascular system, one of which must involve calculating cardiac output. Describe how the tests are performed, what information we can gain from the tests and any limitations to the tests.arrow_forwardHow many electron pairs are shared when a triple bond exists between two carbon atoms? What must he the geometric arrangement around the carbon atoms in a triple bond? Draw the Lewis structure of a simple molecule that contains a triple bond.arrow_forward
- Write the letter of the pair of compounds that illustrates the given isomerism. CH, CH CH3 CH OH D в но H,C. H,C. H,C. CH3 Br CHS OH H F CH Br H3C H3C. CH CH CH3 H3C NH2 CH3 K L AND cis-trans geometric isomerism:Iarrow_forwardIs it possible for alkynes to exhibit cis/trans isomerism? Explain your answer.arrow_forwardNn.200. Subject:- Chemistryarrow_forward
- Answer true or false.Alkenes, alkynes, and arenes are unsaturated hydrocarbons.* Aromatic compounds were so named because many of them have pleasant odors.* According to the resonance model of bonding, benzene is best described as a hybrid of two equivalent contributing structures.* Benzene is a planar molecule.arrow_forward2. To the left, complete the Lewis diagram for a simple amide described by the condensed formula CH(O)NH2. Use your Lewis diagram to fill in the table below: H's Electronic Geometry Shape C O N What bond angles would you assign for this molecule? Hint there should be six different bond angles or five different "types" of bond angles.arrow_forwardplease see attachedarrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





