
Concept explainers
(a)
Interpretation:
The product obtained in the reaction of
Concept introduction:
Answer to Problem 23.44AP
The product obtained in the reaction of
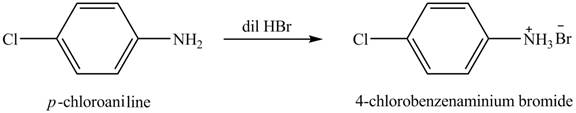
Explanation of Solution
When
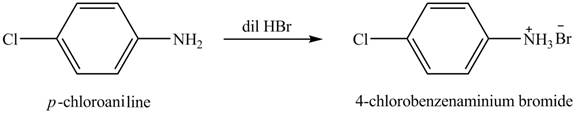
Figure 1
The product obtained in the reaction of
(b)
Interpretation:
The product obtained in the reaction of
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. When alcohol reacts with hydrogen halide it forms
Answer to Problem 23.44AP
The product obtained in the reaction of
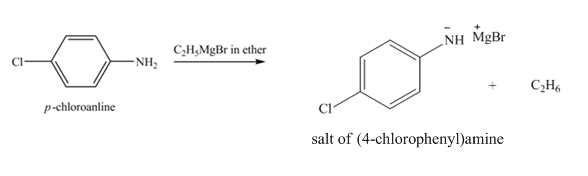
Explanation of Solution
The reaction of

Figure 2
The product obtained in the reaction of
(c)
Interpretation:
The product obtained in the reaction of
Concept introduction:
The formation of diazonium salt from
Answer to Problem 23.44AP
The product obtained in the reaction of
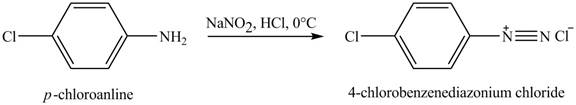
Explanation of Solution
When

Figure 3
The product obtained in the reaction of
(d)
Interpretation:
The product obtained in the reaction of
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. Amines are basic in nature because the nitrogen can donate its lone pairs and also the ability of the nitrogen to accept the proton in water.
Answer to Problem 23.44AP
The product
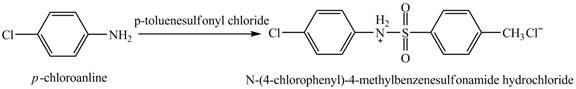
Explanation of Solution
When

Figure 4
The product
(e)
Interpretation:
The product obtained in the reaction of
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. The formation of diazonium salt from aromatic amines takes place using sodium nitrite and hydrochloric acid at low temperatures. Aryl diazonium salts undergo a variety of specific substitution reactions in which the incoming Z group replaces
Answer to Problem 23.44AP
The product
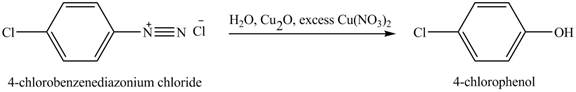
Explanation of Solution
When
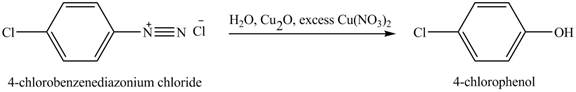
Figure 5
The product obtained in the reaction of
(f)
Interpretation:
The product obtained in the reaction of
Concept introduction:
The formation of diazonium salt from aromatic amines takes place using sodium nitrite and hydrochloric acid at low temperatures. Aryl diazonium salts undergo a variety of specific substitution reactions in which the incoming Z group replaces
Answer to Problem 23.44AP
The product
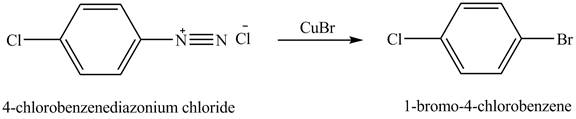
Explanation of Solution
When
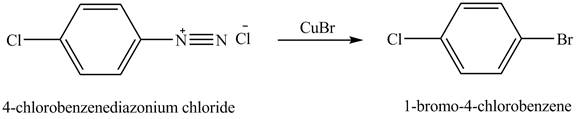
Figure 6
The product
(g)
Interpretation:
The product obtained in the reaction of
Concept introduction:
The formation of diazonium salt from aromatic amines takes place using sodium nitrite and hydrochloric acid at low temperatures. Aryl diazonium salts undergo a variety of specific substitution reactions in which the incoming Z group replaces
Answer to Problem 23.44AP
The product chlorobenzene is obtained in the reaction of the product of part (c) and
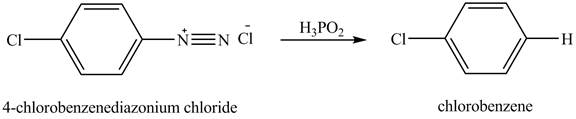
Explanation of Solution
The reduction reaction of
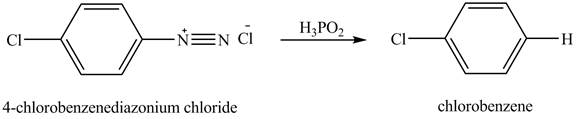
Figure 7
The product chlorobenzene is obtained in the reaction of the product of part (c) and
(h)
Interpretation:
The product obtained in the reaction of
Concept introduction:
The formation of diazonium salt from aromatic amines takes place using sodium nitrite and hydrochloric acid at low temperatures. Aryl diazonium salts undergo a variety of specific substitution reactions in which the incoming Z group replaces
Answer to Problem 23.44AP
The product,
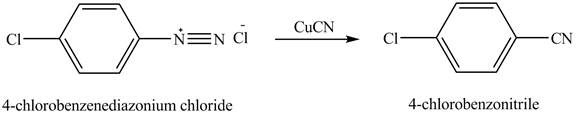
Explanation of Solution
When

Figure 8
The product,
(i)
Interpretation:
The product obtained in the reaction of
Concept introduction:
The formation of diazonium salt from aromatic amines takes place using sodium nitrite and hydrochloric acid at low temperatures. Aryl diazonium salts undergo a variety of specific substitution reactions in which the incoming Z group replaces N2 (a very good leaving group) to form corresponding products.
Answer to Problem 23.44AP
The product
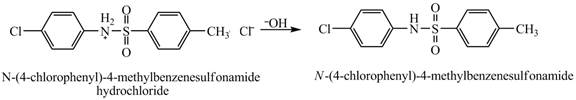
Explanation of Solution
When

Figure 9
The product
Want to see more full solutions like this?
Chapter 23 Solutions
ORGANIC CHEMISTRY SAPLING ACCESS + ETEX
- Following is a synthesis for toremifene, a nonsteroidal estrogen antagonist whose structure is closely related to that of tamoxifen. (a) This synthesis makes use of two blocking groups, the benzyl (Bn) group and the tetrahydropyranyl (THP) group. Draw a structural formula of each group and describe the experimental conditions under which it is attached and removed. (b) Discuss the chemical logic behind the use of each blocking group in this synthesis. (c) Propose a mechanism for the conversion of D to E. (d) Propose a mechanism for the conversion of F to toremifene. (e) Is toremifene chiral? If so, which of the possible stereoisomers are formed in this synthesis?arrow_forwardTreatment of 1-aminoadamantane, C10H17N, with methyl 2,4-dibromobutanoate in the presence of a nonnucleophilic base, R3N, involves two successive SN2 reactions and gives compound A. Propose a structural formula for compound A.arrow_forwardCompound H (C8H6O3) gives a precipitate when treated with hydroxylamine in aqueous ethanol and a silver mirror when treated with Tollens solution. Following is its 1H-NMR spectrum. Deduce the structure of compound H.arrow_forward
- One step in the urea cycle for ridding the body of ammonia is the conversion of argininosuccinate to the amino acid arginine plus fumarate. Propose a mechanism for the reaction, and show the structure of arginine.arrow_forwardCompound I (C11H14O2) is insoluble in water, aqueous acid, and aqueous NaHCO3, but dissolves readily in 10% Na2CO3 and 10% NaOH. When these alkaline solutions are acidified with 10% HCl, compound I is recovered unchanged. Given this information and its 1H-NMR spectrum, deduce the structure of compound I.arrow_forwardWhen cis-2-decalone is dissolved in ether containing a trace of HCl, an equilibrium is established with trans-2-decalone. The latter ketone predominates in the equilibrium mixture. Propose a mechanism for this isomerization and account for the fact that the trans isomer predominates at equilibrium.arrow_forward
- The treatment of (CH3)2C=CHCH2Br with H2O forms B (molecular formula C5H10O) as one of the products. Determine the structure of B from its 1H NMR and IR spectra.arrow_forwardThe compound acetophenone has a very similar molar mass to that of benzoic acid and benzamide. However, acetophenone has a much lower m.p. (20 °C) than both such that, by contrast, it is a liquid at room temperature. By considering intermolecular forces and comparing functional group structure, account for this big difference in physical properties.arrow_forwardSeveral sulfonylureas, a class of compounds containing RSO2NHCONHR, are useful drugs as orally active replacements for injected insulin in patients with adult-onset diabetes. These drugs decrease blood glucose concentrations by stimulating b cells of the pancreas to release insulin and by increasing the sensitivity of insulin receptors in peripheral tissues to insulin stimulation. Tolbutamide is synthesized by the reaction of the sodium salt of p-toluenesulfonamide and ethyl N-butylcarbamate . Propose a mechanism for this step.arrow_forward
- Define the Mechanism - Conversion of Carboxylic Acids to Amides with DCCarrow_forwardWhen A is reacted with hot aqueous NaOH, a compound B of molecular formula C8H11NO is produced. With this information, write the correct structure of B and propose the reaction mechanism (step by step, with the correct use of arrows) to obtain B.arrow_forwardBisphenol A is made on a large scale by a condensation of phenol with acetone. Suggest an appropriate catalyst, and propose a mechanism for this reaction.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

