
(a)
Interpretation:
The product obtained in the reaction of
Concept introduction:
Answer to Problem 23.45AP
The product
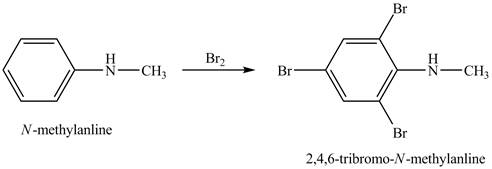
Explanation of Solution
When
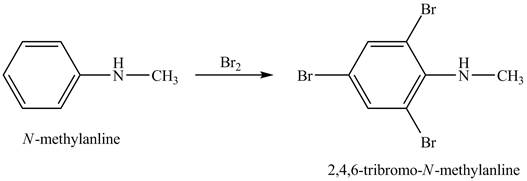
Figure 1
The product obtained in the reaction of
(b)
Interpretation:
The product obtained in the reaction of
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. Amines are basic in nature because the nitrogen can donate its lone pairs and also the ability of the nitrogen to accept the proton in water. The replacement of hydrogen atom attached to a carbon atom of electron-rich benzene ring by an incoming electrophile is known as electrophilic aromatic substitution reaction.
Answer to Problem 23.45AP
The product obtained in the reaction of
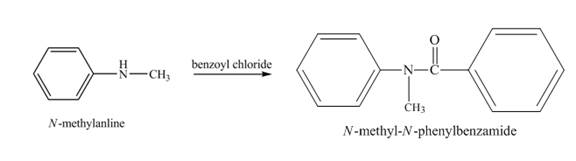
Explanation of Solution
When
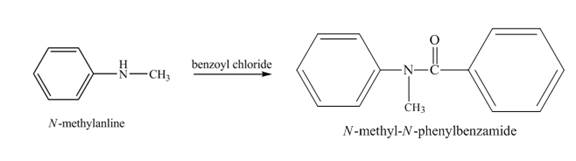
Figure 2
The product obtained in the reaction of
(c)
Interpretation:
The product obtained in the reaction of
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. Amines are basic in nature because the nitrogen can donate its lone pairs and also the ability of the nitrogen to accept the proton in water.
Answer to Problem 23.45AP
The product
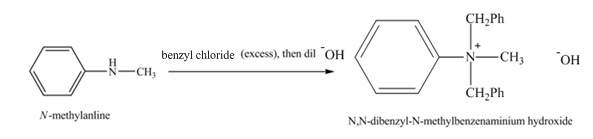
Explanation of Solution
When

Figure 3
The product obtained in the reaction of
(d)
Interpretation:
The product obtained in the reaction of
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. Amines are basic in nature because the nitrogen can donate its lone pairs and also the ability of the nitrogen to accept the proton in water.
Answer to Problem 23.45AP
The product
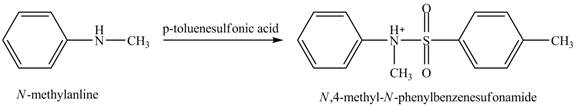
Explanation of Solution
When
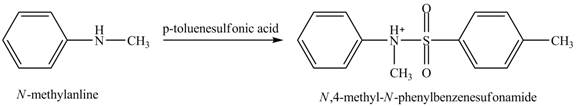
Figure 4
The product obtained in the reaction of
(e)
Interpretation:
The product obtained in the reaction of
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. Amines are basic in nature because the nitrogen can donate its lone pairs and also the ability of the nitrogen to accept the proton in water.
Answer to Problem 23.45AP
The product
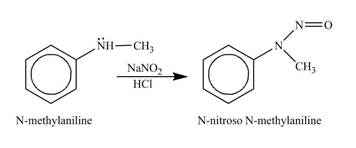
Explanation of Solution
The reagents,
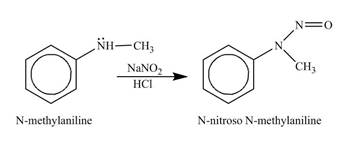
Figure 5
The product obtained in the reaction of
(f)
Interpretation:
The product obtained in the reaction of
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. Hofmann elimination reaction occurs as an anti-elimination reaction. In this reaction, the starting material is quaternary ammonium hydroxide. When quaternary ammonium hydroxide is heated,
Answer to Problem 23.45AP
The product
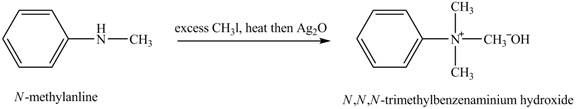
Explanation of Solution
Hofmann elimination reaction occurs as an anti-elimination reaction. In this reaction, the starting material is quaternary ammonium hydroxide. In the given case, when
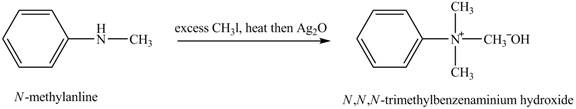
Figure 6
The product obtained in the reaction of
(g)
Interpretation:
The product obtained in the reaction of
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. Amines are basic in nature because the nitrogen can donate its lone pairs and also the ability of the nitrogen to accept the proton in water. Reductive amination reaction is the conversion of the carbonyl group to the amine or it converts one amine to newer amine.
Answer to Problem 23.45AP
The product,
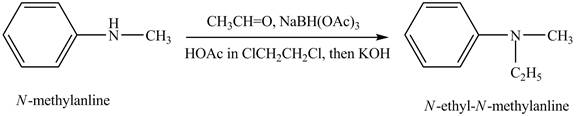
Explanation of Solution
Reductive amination reaction is the conversion of the carbonyl group to the amine or it converts one amine to new amine. In the given case, when
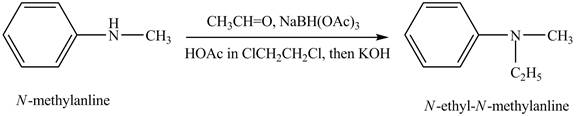
Figure 7
The product obtained in the reaction of
(h)
Interpretation:
The product obtained in the reaction of
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. Amines are basic in nature because the nitrogen can donate its lone pairs and also the ability of the nitrogen to accept the proton in water.
Answer to Problem 23.45AP
The product
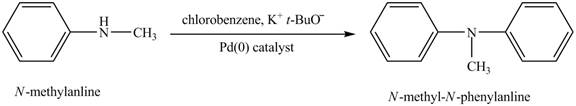
Explanation of Solution
When

Figure 8
The product obtained in the reaction of
Want to see more full solutions like this?
Chapter 23 Solutions
ORGANIC CHEMISTRY (LL)+ SAPLING ACC >BI
- Treatment of 1-aminoadamantane, C10H17N, with methyl 2,4-dibromobutanoate in the presence of a nonnucleophilic base, R3N, involves two successive SN2 reactions and gives compound A. Propose a structural formula for compound A.arrow_forwardOne frequently used method for preparing methyl esters is by reaction of carboxylic acids with diazomethane, CH2N2. The reaction occurs in two steps: (l) protonation of diazomethane by the carboxylic acid to yield methyldiazonium ion, CH3N2+, plus a carboxylate ion; and (2) reaction of the carboxylate ion with CH3N2+. (a) Draw two resonance structures of diazomethane, and account for step 1. (b) What kind of reaction occurs in step 2?arrow_forwardA solution of acetone [(CH3)2C=O] in ethanol (CH3CH2OH) in the presence of a trace of acid was allowed to stand for several days, and a new compound of molecular formula C7H16O2 was formed. The IR spectrum showed only one major peak in the functional group region around 3000 cm−1, and the 1H NMR spectrum is given here. What is the structure of the product?arrow_forward
- Following is a retrosynthetic analysis for an intermediate in the industrial synthesis of vitamin A. (a) Addition of one mole of HCl to isoprene gives 4-chloro-2-methyl-2-butene as the major product. Propose a mechanism for this addition and account for its regioselectivity. (b) Propose a synthesis of the vitamin A precursor from this allylic chloride and ethyl acetoacetate.arrow_forwardFollowing is a synthesis for toremifene, a nonsteroidal estrogen antagonist whose structure is closely related to that of tamoxifen. (a) This synthesis makes use of two blocking groups, the benzyl (Bn) group and the tetrahydropyranyl (THP) group. Draw a structural formula of each group and describe the experimental conditions under which it is attached and removed. (b) Discuss the chemical logic behind the use of each blocking group in this synthesis. (c) Propose a mechanism for the conversion of D to E. (d) Propose a mechanism for the conversion of F to toremifene. (e) Is toremifene chiral? If so, which of the possible stereoisomers are formed in this synthesis?arrow_forwardCarboxylic acids having a second carbonyl group two atoms away lose CO2 (decarboxylate) through an intermediate enolate ion when treated with base. Write the mechanism of this decarboxylation reaction using curved arrows to show the electron flow in each step.arrow_forward
- Give the expected organic product when phenylacetic acid, PhCH2COOH, is treated with reagent Q.)LiAlH4 followed by H2Oarrow_forwardStarting with acetylene and ethylene oxide as the only sources of carbon atoms, show how to prepare the compound Q.)1,6-Hexanediolarrow_forwarda. Compound X is benzene, Y is acetic anhydride acid. Complete the following scheme and determine Z! b. Determine which reagents except acetic acid anhydrides can replace Y!arrow_forward
- The following sequence of steps (R)- 2-octanol to (S)- 2-octanol. Propose structural formulas for intermediates A y B, specify the absolute configuration of each, and account for the inversion of configuration in this sequencearrow_forwardExplain and detail the reaction mechanism: NaBr + H2O + n-butyl alcohol = ?arrow_forwardWrite the reactions of aromatic primary - aliphatic secondary and tertiary amines with 2 N HCl and 5% NaNO2 in an ice bath at 5°C.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


