
Concept explainers
(a)
Interpretation:
The structure of N-isopropylaniline is to be drawn.
Concept introduction:
Answer to Problem 23.1P
The structure of N-isopropylaniline is shown below.
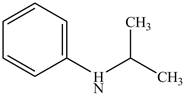
Explanation of Solution
The name N-isopropylaniline indicates that it consists of the structure of aniline compound. One hydrogen atom of
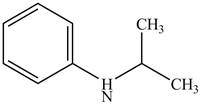
Figure 1
The structure of N-isopropylaniline is shown in Figure 1.
(b)
Interpretation:
The structure of tert-butylamine is to be drawn.
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia. Amines are basic in nature because the nitrogen can donate its lone pairs and also the ability of the nitrogen to accept the proton in water.
Answer to Problem 23.1P
The structure of tert-butylamine is shown below.
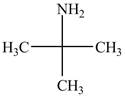
Explanation of Solution
The name tert-butylamine indicates that it consist of tert-butyl group. The tert-butyl group is attached to an amine group. The structure of tert-butylamine is shown below.
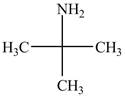
Figure 2
The structure of tert-butylamine is shown in Figure 2.
(c)
Interpretation:
The structure of
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia. Amines are basic in nature because the nitrogen can donate its lone pairs and also the ability of the nitrogen to accept the proton in water.
Answer to Problem 23.1P
The structure of
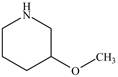
Explanation of Solution
The name
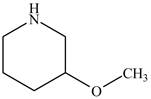
Figure 3
The structure of
(d)
Interpretation:
The structure of
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia. Amines are basic in nature because the nitrogen can donate its lone pairs and also the ability of the nitrogen to accept the proton in water.
Answer to Problem 23.1P
The structure of
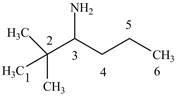
Explanation of Solution
The name
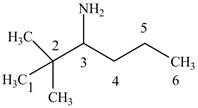
Figure 4
The structure of
(e)
Interpretation:
The structure of
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia. Amines are basic in nature because the nitrogen can donate its lone pairs and also the ability of the nitrogen to accept the proton in water.
Answer to Problem 23.1P
The structure of
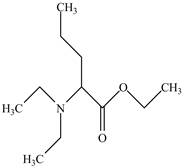
Explanation of Solution
The name
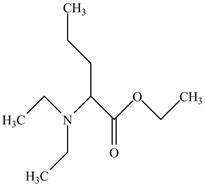
Figure 5
The structure of
(f)
Interpretation:
The structure of
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia. Amines are basic in nature because the nitrogen can donate its lone pairs and also the ability of the nitrogen to accept the proton in water.
Answer to Problem 23.1P
The structure of
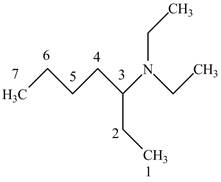
Explanation of Solution
The name
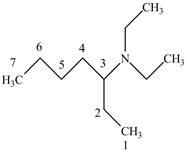
Figure 6
The structure of
Want to see more full solutions like this?
Chapter 23 Solutions
ORGANIC CHEMISTRY (LL)+ SAPLING ACC >BI
- Several compounds have been found to inhibit -lactamase, and drugs based on these compounds can be taken in combination with penicillins and cephalosporins to restore their effectiveness when resistance is known to be a problem. The commonly prescribed formulation called Augmentin is a combination of the -lactamase inhibitor shown below with amoxicillin (shown above). It is used to treat childhood ear infections when resistance is suspected, and many kids know it as the white liquid that tastes like bananas. Which of the statements below are true statements? 1. The stereochemistry of the fusion between the four-and five-membered rings in the inhibitor and amoxicillin are different. 2. The inhibitor possesses enol ether and allylic alcohol functional groups while the antibiotic possesses a phenol and a secondary amide functional group. 3. Neither the inhibitor nor the antibiotic contains strained rings. 4. Both 1 and 2 are true.arrow_forwardComplete each of the following by supplying the missing reagents. Draw the structures of each of the reactants and products. a. N-Methylpropanamide + ? --> propanoic acid + ? b. N,N-Dimethylacetamide + strong acid --> ? + ? c. Formamide + strong acid --> ? + ?arrow_forwardDescribe the reaction involving the preparation of N-Propylcycloheptanamine. State the kind of reaction.arrow_forward
- Comment on the solubilities of the following amines in water. Please write soluble or insoluble next to the compound. 1. Cyclohexylamine - 2. Aniline - 3. Pyridine - 4. Triethylamine -arrow_forwardWhich of these 3 compounds is the least basic: aniline, 2-propylamine and 1-(2-aminophenyl)ethenone.arrow_forwardThe hydrolysis of an amide in acidic conditions forms A. a carboxylate salt and an alcohol B. a carboxylate salt and an amine C. an alcohol and an amine salt (an ammonium ion) D. a carboxylic acid and an amine salt (an ammonium ion)arrow_forward
- Explain why a secondary amine forms a nitrosamine rather than a diazonium ion when it reacts with anitrosonium ion.arrow_forwardIf a quaternary ammonium ion can undergo an elimination reaction with a strong base, why can’t a protonated tertiary amine undergo the same reaction?arrow_forwardripelennamine is a histamine H1 antagonist with low sedative action but frequent gastrointestinal irritation. It is used to treat asthma; hay fever; urticaria; and rhinitis. Based on biotransformations discussed in class, draw the structures of 3 possible metabolites of tripelennamine.arrow_forward
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


