
Concept explainers
(a)
Interpretation:
The increasing order of basicity of propylamine, ammonia, and dipropylamine in aqueous solution is to be arranged.
Concept introduction:
Answer to Problem 23.7P
The increasing order of basicity of propylamine, ammonia, and dipropylamine in aqueous solution is shown below.
Explanation of Solution
It is known that the order of basicity of amines in aqueous solution is
The increasing order of basicity of propylamine, ammonia, and dipropylamine in aqueous solution is shown below.
(b)
Interpretation:
The increasing order of basicity of
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia. Amines are basic in nature because the nitrogen can donate its lone pairs and also the ability of the nitrogen to accept the proton in water.
Answer to Problem 23.7P
The increasing order of basicity of
Explanation of Solution
It is known that the order of basicity of amines in aqueous solution solution is
The increasing order of basicity of
(c)
Interpretation:
The increasing order of basicity of aniline, methyl m-aminobenzoate, and methyl p-aminobenzoate in aqueous solution is to be arranged.
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia. Amines are basic in nature because the nitrogen can donate its lone pairs and also the ability of the nitrogen to accept the proton in water.
Answer to Problem 23.7P
The increasing order of basicity of aniline, methyl m-aminobenzoate, and methyl p-aminobenzoate in aqueous solution is shown below.
Explanation of Solution
The structure of aniline, methyl m-aminobenzoate, and methyl p-aminobenzoate is shown below.
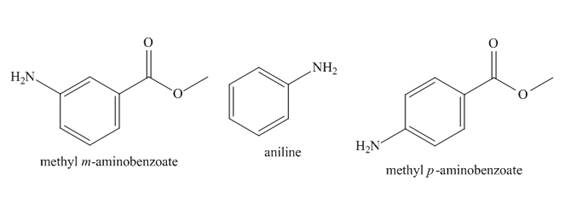
Figure 1
Amines are basic in nature because the nitrogen can donate its lone pairs and also the ability of the nitrogen to accept the proton in water.
The basicity of aniline substituted compound depends on the group that is attached to benzene ring. If the group attached to benzene ring is electron withdrawing group, it will decrease the basicity of the compound. If the group attached to benzene ring is electron donating group, it will increase the basicity of the compound.
There is no group attached in case of aniline. Therefore, it is most basic. In case of methyl m-aminobenzoate and methyl p-aminobenzoate, the
The increasing order of basicity of aniline, methyl m-aminobenzoate, and methyl p-aminobenzoate in aqueous solution is shown below.
(d)
Interpretation:
The increasing order of basicity of benzylamine, p-nitrobenzylamine, cyclohexylamine, aniline in aqueous solution is to be arranged.
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia. Amines are basic in nature because the nitrogen can donate its lone pairs and also the ability of the nitrogen to accept the proton in water.
Answer to Problem 23.7P
The increasing order of basicity of benzylamine, p-nitrobenzylamine, cyclohexylamine, aniline in aqueous solution is shown below.
Explanation of Solution
The structure of benzylamine, p-nitrobenzylamine, cyclohexylamine, aniline is shown below.
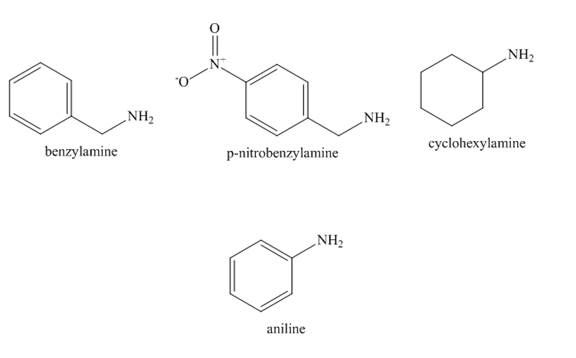
Figure 2
Amines are basic in nature because the nitrogen can donate its lone pairs and also the ability of the nitrogen to accept the proton in water.
In case of aniline, the lone pair present at nitrogen atom is resonate with the benzene ring. Therefore, the electron density on nitrogen is decreased. Therefore, it is least basic.
In case of cyclohexylamine, there is no resonance. The cyclohexyl group is an electron donating group which increases the electron density on nitrogen atom. Therefore, the basicity of cyclohexylamine increased.
In case of benzylamine there is also no resonance because
In case of p-nitrobenzylamine there is also no resonance because
The increasing order of basicity of benzylamine, p-nitrobenzylamine, cyclohexylamine, aniline in aqueous solution is shown below.
Want to see more full solutions like this?
Chapter 23 Solutions
EBK ORGANIC CHEMISTRY STUDY GUIDE AND S
- Histamine, whose release in the body triggers nasal secretions and constricted airways, has three nitrogen atoms. List them in order of increasing basicity and explain your ordering.arrow_forwardi) What property of amines is responsible for their being basic ii) Arrange the following compounds in order of increasing basic strength putting the least basic first. NH3 CH,CONH; O NH2 CH,CH;NH; D C. c) What general name is given to the reaction between phenylamine and nitrous acid at 5°C ii) Write the equation for the reaction in c(i). + Ajouter une légende.. > Statut (Personnalisé)arrow_forwardRank the Acidity of the following sets of compounds. Give an explanation for the answers.arrow_forward
- Rank each set of compounds in order of increasing basicity.(a) NaOH, NH3, CH3NH2, Ph¬NH2 (arrow_forwardArrange the members of each group in order of decreasing basicity: (a) Ammonia, aniline, methylamine (b) Acetanilide, aniline, N-methylaniline (c) 2,4-Dichloroaniline, 2,4-dimethylaniline, 2,4-dinitroaniline (d) 3,4-Dichloroaniline, 4-chloro-2-nitroaniline, 4-chloro-3-nitroaniline (e) Dimethylamine, diphenylamine, N-methylanilinearrow_forwardArrange the following molecules in increasing order of acidity. Base it only on their structural differences and explain how it is so. 1. HF, CH3CH2CH2OH, CH3CH2COOH 2. Ethyl amine, Ethanol, Propanearrow_forward
- Carboxylic Acid Derivatives Resolve by synthesis, step by step.arrow_forwardPropyl amine can be prepared from the reaction of Ammonia and 1 point Propyi bromide Propanone Propane Propene The typels of amines that exhibit a degree of acidity due to the presence * point of protic Hydrogen islare: (a) primary amine, (b) secondary amine, (c) tertiary amine, (d) quaternary amine a a and b b and c c and d Amines are basic in nature because of the presence of* 1 point its polarity its trigonal pyramidal geometry lone pair in N N-H bondarrow_forwardHypoglycin A, an amino acid derivative found in unripened lychee, is anacutely toxic compound that produces seizures, coma, and sometimesdeath in undernourished children when ingested on an empty stomach. (a) Draw the neutral, positively charged, and negatively charged forms of hypoglycin A. (b) Which form predominates at pH = 1, 6, and 11? (c) What is the structure of hypoclycin A at its isoelectric point?arrow_forward
- Arrange each group of compounds in order of increasing basicity.(a) CH3COO-, ClCH2COO-, and PhO- (b) sodium acetylide, sodium amide, and sodium acetate(c) sodium benzoate, sodium ethoxide, and sodium phenoxide (d) pyridine, sodium ethoxide, and sodium acetatearrow_forward3. Rank the following primary amines (anilines in this case) in order of increasing basicity (1 = least basic, 4 = most basic). Write the conjugate acid and provide a brief explanation for your ordering. NH2 NH2 NH2 NH2 OCH3 NO2 CH3 A В C Darrow_forwardArrange the following in increasing order of boiling point. Give reason. Methylamine, trimethylamine, dimethylamine Arrange the following in increasing order of basicity. Give reason. Methylamine, trimethylamine, dimethylamine Arrange the following in increasing order of solubility in H2O. Give reason. Methylamine, trimethylamine, dimethylaminearrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


