
Concept explainers
Name the following
(a) CH3CH2CH2NH2
(b) (CH3)3N
(c) (CH3)(C2H5)NH
(d) C6H13NH2
(a)
Interpretation: The name of the following amine has to be written.
Concept introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
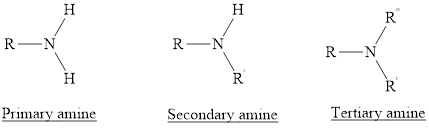
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Answer to Problem 48PS
The systematic name of the given amine,
Explanation of Solution
The molecular formula of the given amine is
It is a primary amine
One propyl
Therefore, the name of the given amine is propylamine.
(b)
Interpretation: The name of the following amine has to be written.
Concept introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Answer to Problem 48PS
The systematic name of the given amine,
Explanation of Solution
The molecular formula of the given amine is
Three methyl
It is a tertiary amine
Therefore,
The name of the given amine is N,N-trimethylamine.
(c)
Interpretation: The name of the following amine has to be written.
Concept introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Answer to Problem 48PS
The systematic name of the given amine,
Explanation of Solution
The molecular formula of the given amine is
One methyl
It is a secondary amine
Therefore, the name of the given amine is N-ethylmethylamine.
(d)
Interpretation: The name of the following amine has to be written.
Concept introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Answer to Problem 48PS
The systematic name of the given amine,
Explanation of Solution
The molecular formula of the given amine is
It is a primary amine
One hexyl
Therefore the name of the given amine is hexylamine.
Want to see more full solutions like this?
Chapter 23 Solutions
CHEMISTRY+CHEM.REACT. (LL)-W/OWL ACCESS
- 1,4-Diazabicyclo[2.2.2]octane (abbreviated DABCO) is a tertiary amine that catalyzes transesterification reactions. Explain how it does this.arrow_forwardPropose structures for amines that fit these descriptions:(a) A secondary amine with formula C5H13N(b) A tertiary amine with formula C6H13N(c) A cyclic quaternary amine that has the formula C6H14N+arrow_forwardWrite structural formulas for all the amines of molecular formula C4H11N. Give an acceptable name for each one, and classify it as a primary, secondary, or tertiary amine.arrow_forward
- In addition to HCl, what is the product of the reaction of aprimary amine with an acid chloride? Draw the structureof that product and describe its featuresarrow_forward(a) Compound Z is a tertiary aromatic amine with the formula, C8H11N. Provide a chemical structure for compound Z. (b)nDraw the structure of the product formed exclusively when nitrous acid reacts with Z.arrow_forward3 Compare the basicity of amines with other common bases, and explain how theirbasicity varies with hybridization and aromaticity.arrow_forward
- Considering Hinsberg's Method to determine, 1°, 2°, 3° amines. Draw the structure of the products of the following reactions. Aniline (1°) + BSC, excess NaOH(DRAW STRUCTURE), then, + HCl (DRAW STRUCTURE) Diethylamine (2°)+ BSC, excess NaOH (DRAW STRUCTURE), then, + HCl (DRAW STRUCTURE) Triethylamine (3°)+ BSC, excess NaOH(DRAW STRUCTURE), then, + HCl (DRAW STRUCTURE)arrow_forwardAssume for the purposes of this problem that to be an alcohol (-ol) or an amine (-amine), the hydroxyl or amino group must be bonded to a tetrahedral (sp3 hybridized) carbon atom. Write the structural formula of a compound with an unbranched chain of four carbon atoms that is an: Q. Alkenaminearrow_forwardGive a suitable chemical reaction to distinguish between a primary and a secondary amine. Suggest one way of Increasing the basicity of an amine and give a specific example of its application.arrow_forward
- Explain amine catalysis in urethane bond formationarrow_forwardPredict the major products formed when the following amines undergo exhaustivemethylation, treatment with Ag2O, and heating.2-methylpiperidinearrow_forwardA chiral amine A having the R conguration undergoes Hofmann elimination to form an alkene B as the major product. B is oxidatively cleaved with ozone, followed by CH3SCH3, to form CH2 = O and CH3CH2CH2CHO. What are the structures of A and B?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


