
Chemistry: Atoms First
2nd Edition
ISBN: 9780077646479
Author: Burdge
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 23.3, Problem 23.3.3SR
Which of the following pairs of species are resonance structures?
(a) 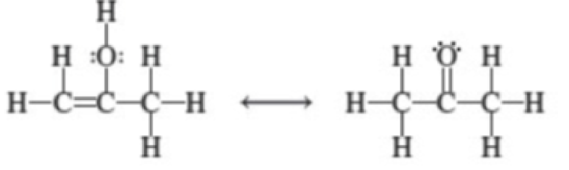
(b) 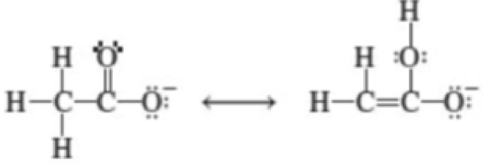
(c) 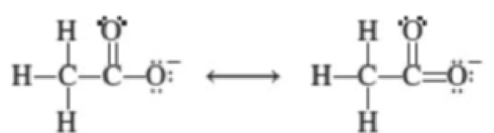
(d) 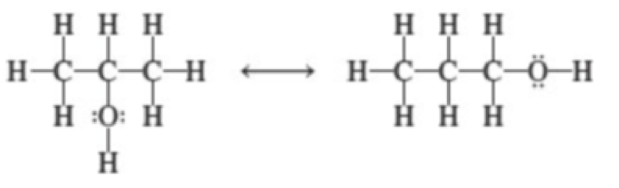
(e) 
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
Chemistry: Atoms First
Ch. 23.2 - Prob. 23.1WECh. 23.2 - Prob. 1PPACh. 23.2 - Prob. 1PPBCh. 23.2 - Prob. 1PPCCh. 23.2 - Prob. 23.2WECh. 23.2 - Give the systematic IUPAC name for each of the...Ch. 23.2 - Prob. 2PPBCh. 23.2 - Prob. 2PPCCh. 23.2 - Prob. 23.2.1SRCh. 23.2 - Prob. 23.2.2SR
Ch. 23.2 - Prob. 23.2.3SRCh. 23.2 - Prob. 23.2.4SRCh. 23.2 - Prob. 23.2.5SRCh. 23.2 - Prob. 23.2.6SRCh. 23.3 - Prob. 23.3WECh. 23.3 - Prob. 3PPACh. 23.3 - Prob. 3PPBCh. 23.3 - Prob. 3PPCCh. 23.3 - Prob. 23.4WECh. 23.3 - Prob. 4PPACh. 23.3 - Prob. 4PPBCh. 23.3 - Prob. 4PPCCh. 23.3 - Prob. 23.3.1SRCh. 23.3 - Prob. 23.3.2SRCh. 23.3 - Which of the following pairs of species are...Ch. 23.3 - Prob. 23.3.4SRCh. 23.5 - Prob. 23.5WECh. 23.5 - Prob. 5PPACh. 23.5 - Prob. 5PPBCh. 23.5 - Prob. 23.5.1SRCh. 23.5 - Prob. 23.5.2SRCh. 23 - Prob. 23.1QPCh. 23 - Prob. 23.2QPCh. 23 - Prob. 23.3QPCh. 23 - Prob. 23.4QPCh. 23 - Prob. 23.5QPCh. 23 - Prob. 23.6QPCh. 23 - Prob. 23.7QPCh. 23 - Prob. 23.8QPCh. 23 - Prob. 23.9QPCh. 23 - Name each of the following compounds.Ch. 23 - Prob. 23.11QPCh. 23 - Prob. 23.12QPCh. 23 - Prob. 23.13QPCh. 23 - Prob. 23.14QPCh. 23 - Prob. 23.15QPCh. 23 - Prob. 23.16QPCh. 23 - Prob. 23.17QPCh. 23 - Prob. 23.18QPCh. 23 - Prob. 23.19QPCh. 23 - Prob. 23.20QPCh. 23 - Prob. 23.21QPCh. 23 - Prob. 23.22QPCh. 23 - Prob. 23.23QPCh. 23 - Prob. 23.24QPCh. 23 - Prob. 23.25QPCh. 23 - Prob. 23.26QPCh. 23 - Prob. 23.27QPCh. 23 - Prob. 23.28QPCh. 23 - Prob. 23.29QPCh. 23 - Prob. 23.30QPCh. 23 - Prob. 23.31QPCh. 23 - Prob. 23.32QPCh. 23 - Prob. 23.33QPCh. 23 - Prob. 23.34QPCh. 23 - Fill in the blanks in the given paragraph with the...Ch. 23 - Prob. 23.36QPCh. 23 - Draw all possible structural isomers for the...Ch. 23 - Prob. 23.38QPCh. 23 - Prob. 23.39QPCh. 23 - Prob. 23.40QPCh. 23 - Prob. 23.41QPCh. 23 - Prob. 23.42QPCh. 23 - Prob. 23.43QPCh. 23 - Prob. 23.44QPCh. 23 - Prob. 23.45QPCh. 23 - Prob. 23.46QPCh. 23 - Prob. 23.47QPCh. 23 - Prob. 23.48QPCh. 23 - Prob. 23.49QPCh. 23 - Prob. 23.50QPCh. 23 - Prob. 23.51QPCh. 23 - Prob. 23.52QPCh. 23 - (a) Define carbocation. (b) Which of the following...Ch. 23 - Prob. 23.54QPCh. 23 - Prob. 23.55QPCh. 23 - Prob. 23.56QPCh. 23 - Prob. 23.57QPCh. 23 - Prob. 23.58QPCh. 23 - Prob. 23.59QPCh. 23 - Consider the following reactions of butanal. In...Ch. 23 - Prob. 23.61QPCh. 23 - Prob. 23.62QPCh. 23 - Prob. 23.63QPCh. 23 - Prob. 23.64QPCh. 23 - Prob. 23.65QPCh. 23 - Prob. 23.66QPCh. 23 - Prob. 23.67QPCh. 23 - Prob. 23.68QPCh. 23 - Prob. 23.69QPCh. 23 - Prob. 23.70QPCh. 23 - Prob. 23.71QPCh. 23 - Prob. 23.72QPCh. 23 - Prob. 23.73QPCh. 23 - Prob. 23.74QPCh. 23 - Prob. 23.75QPCh. 23 - Prob. 23.76QPCh. 23 - Prob. 23.77QPCh. 23 - Prob. 23.78QPCh. 23 - Prob. 23.79QPCh. 23 - Prob. 23.80QPCh. 23 - Prob. 23.81QPCh. 23 - Prob. 23.82QPCh. 23 - Prob. 23.83QPCh. 23 - Prob. 23.84QPCh. 23 - Prob. 23.85QPCh. 23 - Prob. 23.86QPCh. 23 - Prob. 23.87QPCh. 23 - Prob. 23.88QPCh. 23 - Prob. 23.89QPCh. 23 - Prob. 23.90QPCh. 23 - Prob. 23.91QPCh. 23 - Prob. 23.92QPCh. 23 - Prob. 23.93QPCh. 23 - Prob. 23.94QPCh. 23 - Prob. 23.95QPCh. 23 - Prob. 23.96QPCh. 23 - Prob. 23.97QPCh. 23 - Prob. 23.98QPCh. 23 - Prob. 23.99QPCh. 23 - Prob. 23.100QPCh. 23 - Prob. 23.101QPCh. 23 - Prob. 23.102QPCh. 23 - Prob. 23.103QPCh. 23 - Prob. 23.104QPCh. 23 - Prob. 23.105QP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Which of the following solutions has the higher molarity? 10 ppm KI in water or 10,000 ppb KBr in water 0.25 ma...
CHEMISTRY-TEXT
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living by Chemistry
Fully developed conditions are known to exist for water flowing through a 25-nim-diameer tube at 0.01 kg/s and ...
Fundamentals of Heat and Mass Transfer
Write the electron configurations far each of the following elements: (a) Sc. (b) Ti. (c) Cr. (d) Fe. (e) Ru
Chemistry by OpenStax (2015-05-04)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Aspirin is made from salicylic acid, which has this Lewis structure: (a) Which is the longest carbon-carbon bond? (b) Which is the strongest carbon-oxygen bond? (c) Draw resonance structures for this molecule.arrow_forwardGiven the bonds C N, C H, C Br, and S O, (a) which atom in each is the more electronegative? (b) which of these bonds is the most polar?arrow_forwardWhat aspect of the following Lewis structure indicates that the concept of coordinate covalency is needed to explain the bonding in the molecule?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY