
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
8th Edition
ISBN: 9780134015187
Author: John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 23.4, Problem 23.10P
Interpretation Introduction
Interpretation:
The structure of the micelle in a saturated fatty acid salt has to be drawn and their parts need to be shown.
Concept Introduction:
Micelle formation:
A sphere-shaped cluster formed when the accumulation of soap or detergent molecules so that their hydrophilic ends are on the surface and hydrophobic ends is in the centre are formed
Acids have two different parts
- (1) Hydrophilic (water-loving),
- (2) Hydrophobic (water-fearing).
Hydrophilic (water-loving):
The compound has the carboxyl group (
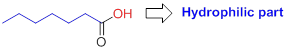
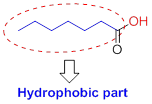
Hydrophobic (water-fearing):
The compound has alkyl chain group is called as hydrophilic.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Match the area of dipeptide with the number
Describe the properties of the cornstarch mixture. Would you call it solid or liquid? Explain
What could be the reason why hydroxylation of proline residues is critical to the structural integrity of collagen?
Select the correct response
The hydroxyl moities in Pro form disulfide bonds which stabilize collagen structure.
The hydroxyl moities in Pro form ionic bonds (salt bridges) which stabilize collagen structure.
The hydroxyl moities in Pro form hydrophobic interactions which stabilize collagen structure.
The hydroxyl moities in Pro form peptide bonds which stabilize collagen structure.
The hydroxyl moities in Pro form H-bonds which stabilize collagen structure.
Chapter 23 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
Ch. 23.1 - Use Figure 23.1 to identify the family of lipids...Ch. 23.2 - Prob. 23.2PCh. 23.2 - Prob. 23.3PCh. 23.2 - Prob. 23.4KCPCh. 23.3 - Prob. 23.1CIAPCh. 23.3 - Prob. 23.2CIAPCh. 23.3 - Prob. 23.3CIAPCh. 23.3 - Prob. 23.5PCh. 23.3 - Prob. 23.6PCh. 23.3 - Prob. 23.7KCP
Ch. 23.4 - Prob. 23.8PCh. 23.4 - Prob. 23.9PCh. 23.4 - Prob. 23.10PCh. 23.4 - Prob. 23.11PCh. 23.5 - Prob. 23.12PCh. 23.5 - Draw the structure of the sphingomyelin that...Ch. 23.5 - Draw the structure of the glycerophospholipid that...Ch. 23.5 - Prob. 23.16PCh. 23.7 - Prob. 23.17KCPCh. 23.7 - Prob. 23.4CIAPCh. 23.7 - Prob. 23.6CIAPCh. 23.7 - Prob. 23.7CIAPCh. 23.7 - Prob. 23.8CIAPCh. 23.7 - Prob. 23.18PCh. 23.7 - Prob. 23.19PCh. 23.7 - Prob. 23.20KCPCh. 23 - The fatty acid composition of three...Ch. 23 - Prob. 23.23UKCCh. 23 - According to the fluid-mosaic model (Figure 23.7),...Ch. 23 - Dipalmitoylphosphatidylcholine (DPPC) is a...Ch. 23 - Prob. 23.26APCh. 23 - Prob. 23.27APCh. 23 - Prob. 23.28APCh. 23 - Prob. 23.29APCh. 23 - Differentiate between saturated, monounsaturated,...Ch. 23 - Are the carboncarbon double bonds in naturally...Ch. 23 - Prob. 23.32APCh. 23 - Prob. 23.33APCh. 23 - Which of these fatty acids has the lower melting...Ch. 23 - Which of these fatty acids has the higher melting...Ch. 23 - Prob. 23.36APCh. 23 - Prob. 23.37APCh. 23 - Prob. 23.38APCh. 23 - Prob. 23.39APCh. 23 - What function does a wax serve in a plant or...Ch. 23 - Prob. 23.41APCh. 23 - Prob. 23.42APCh. 23 - What kind of lipid is spermacetia fat, a wax, or a...Ch. 23 - Prob. 23.44APCh. 23 - Prob. 23.45APCh. 23 - Prob. 23.46APCh. 23 - Prob. 23.47APCh. 23 - Prob. 23.48APCh. 23 - Prob. 23.50APCh. 23 - Prob. 23.52APCh. 23 - Prob. 23.53APCh. 23 - Describe the difference between a triacylglycerol...Ch. 23 - Why are glycerophospholipids, rather than...Ch. 23 - Prob. 23.56APCh. 23 - Prob. 23.57APCh. 23 - Why are glycerophospholipids more soluble in water...Ch. 23 - Prob. 23.59APCh. 23 - Prob. 23.60APCh. 23 - Prob. 23.61APCh. 23 - Draw the structure of a glycerophospholipid that...Ch. 23 - Prob. 23.63APCh. 23 - What is a major function of cholesterol in your...Ch. 23 - Prob. 23.65APCh. 23 - Prob. 23.66APCh. 23 - Prob. 23.67APCh. 23 - Explain how a micelle differs from a membrane...Ch. 23 - Prob. 23.69APCh. 23 - Prob. 23.70APCh. 23 - Prob. 23.71APCh. 23 - Prob. 23.72APCh. 23 - Prob. 23.73APCh. 23 - Prob. 23.74APCh. 23 - Prob. 23.75APCh. 23 - Draw the structure of a triacylglycerol made from...Ch. 23 - Prob. 23.79CPCh. 23 - Prob. 23.80CPCh. 23 - Explain why cholesterol is not saponifiable.Ch. 23 - Draw cholesterol acetate. Is this molecule...Ch. 23 - Prob. 23.83CPCh. 23 - Prob. 23.84CPCh. 23 - Prob. 23.85CPCh. 23 - Prob. 23.86CPCh. 23 - Prob. 23.88GP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- What will be happen if you mix; 1ml of 5% solution of albumin and 5 mL of 95% ethanol 1ml of 5% solution of gelatin and 5 mL of 95% ethanol 1ml of 5% solution of peptone and 5 mL of 95% ethanol Mix and observe the formation of precipitate.arrow_forwardWhat is acetic orcein and what structure does it stain?arrow_forwardIn the preparation of sauces that involve mixing water and melted butter, egg yolks are added to prevent separation. How do egg yolks prevent separation? Hint: Egg yolks are rich in phosphatidyl choline (lecithin).arrow_forward
- Did the slime pick up water soluble or water insoluble inks? From these results, what can you conclude about the polarity of slime molecules?arrow_forwardDraw the structure of ethanolamine sphingomyelin formed from linoleic acid Draw the structure of serine sphingomyelin formed from arachidic acidarrow_forwardWhy are viens blue?arrow_forward
- What is the chemical composition of peanut, peanut shell, and corn grain?arrow_forwardWhich contains more protein (C—N bonds), egg albumen or honey? How can you tell?arrow_forwardWould you expect albumin to have a higher or lower proportion of free amino groups to free carboxyl groups than casein? Casein which is more soluble in acid, will it have more amino groups or more carboxyl groups?arrow_forward
- In the structure in the picture, the monosaccharide units (alpha-D-galactopyranosyl and beta-D-allopyranose) are linked via alpha-1->3 glycosidic bond. Then, the formed disaccharide units are linked via beta-1->4 glycosidic bond. Then, an Oligosaccharide is formed which has 10 monosaccharide units, meaning that it contains 5 disaccharide units. Question: Is this oligosaccharide a good substrate for glycolysis? Provide two reasons for your answer.arrow_forwardWhat molecules are present in regions of gel corresponding to A? What happens upon heatshock? What does Band B represent?arrow_forwardWhen betadine is dropped into a ripe banana, does this turn into orange/brown or blue-black? Does a ripe banana contain starch? Take not that unripe and ripe banana has different results.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
