
Bundle: Organic Chemistry, Loose-leaf Version, 9th + LMS Integrated for OWLv2, 4 terms (24 months) Printed Access Card
9th Edition
ISBN: 9781305780170
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 23.SE, Problem 33MP
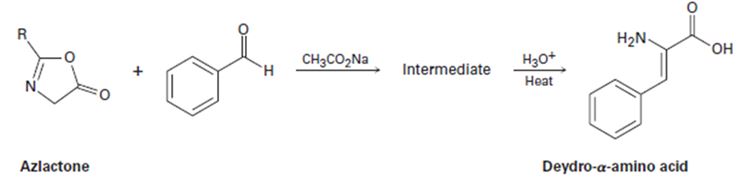
Azlactones are important starting materials used in the synthesis of dehydro α-aminoacids. They react with
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Define the Mechanism - Conversion of Carboxylic Acids to Amides with DCC
Ethanolamine ammonia lyase, a coenzyme B12–requiring enzyme, catalyzes the following reaction. Propose a mechanism for this reaction.
Treatment of pentanedioic (glutaric) anhydride with ammonia at elevated temperature leads to a compound of molecular formula C5H7NO2. What is the structure of this product? [Hint: You need to think about the reactivity not only of acid anhydrides but also of amides and carboxylic acids]
Chapter 23 Solutions
Bundle: Organic Chemistry, Loose-leaf Version, 9th + LMS Integrated for OWLv2, 4 terms (24 months) Printed Access Card
Ch. 23.1 - Prob. 1PCh. 23.1 - Prob. 2PCh. 23.3 - Prob. 3PCh. 23.3 - Prob. 4PCh. 23.4 - Prob. 5PCh. 23.4 - 1-Butanol is prepared commercially by a route that...Ch. 23.4 - Prob. 7PCh. 23.5 - Prob. 8PCh. 23.6 - Prob. 9PCh. 23.6 - What product would you Expect to obtain from base...
Ch. 23.7 - Show the products you would expect to obtain by...Ch. 23.7 - Prob. 12PCh. 23.8 - What product would you expect from the following...Ch. 23.9 - What product would you expect From the following...Ch. 23.9 - Prob. 15PCh. 23.10 - Prob. 16PCh. 23.10 - Prob. 17PCh. 23.10 - Prob. 18PCh. 23.11 - Prob. 19PCh. 23.11 - Show how you might use an enamine reaction to...Ch. 23.12 - Prob. 21PCh. 23.12 - How would you prepare the following compound using...Ch. 23.SE - Prob. 23VCCh. 23.SE - Prob. 24VCCh. 23.SE - Prob. 25VCCh. 23.SE - The following molecule was formed by a Robinson...Ch. 23.SE - Prob. 27MPCh. 23.SE - Prob. 28MPCh. 23.SE - Predict the product(s) and provide the mechanism...Ch. 23.SE - Predict the product(s) and provide the mechanism...Ch. 23.SE - Predict the product(s) and provide the mechanism...Ch. 23.SE - Knoevenagel condensation is a reaction involving...Ch. 23.SE - Azlactones are important starting materials used...Ch. 23.SE - Prob. 34MPCh. 23.SE - Isoleucine, another of the twenty amino acids...Ch. 23.SE - The first step in the citric acid cycle of food...Ch. 23.SE - Prob. 37MPCh. 23.SE - The Knoevenagel reaction is a carbonyl...Ch. 23.SE - The Darzens reaction invoIves a two-step,...Ch. 23.SE - The following reaction involves a hydrolysis...Ch. 23.SE - Prob. 41MPCh. 23.SE - Prob. 42MPCh. 23.SE - Prob. 43MPCh. 23.SE - Propose a mechanism for the following...Ch. 23.SE - Prob. 45MPCh. 23.SE - Prob. 46MPCh. 23.SE - Prob. 47MPCh. 23.SE - Prob. 48APCh. 23.SE - Prob. 49APCh. 23.SE - Prob. 50APCh. 23.SE - Prob. 51APCh. 23.SE - Prob. 52APCh. 23.SE - Prob. 53APCh. 23.SE - Prob. 54APCh. 23.SE - Prob. 55APCh. 23.SE - Prob. 56APCh. 23.SE - Prob. 57APCh. 23.SE - Prob. 58APCh. 23.SE - In the mixed Claisen reaction of cyclopentanone...Ch. 23.SE - Ethyl dimethylacetoacetate reacts instantly at...Ch. 23.SE - Prob. 61APCh. 23.SE - Prob. 62APCh. 23.SE - The so-called Wieland-Miescher ketone is a...Ch. 23.SE - Prob. 64APCh. 23.SE - Prob. 65APCh. 23.SE - Prob. 66APCh. 23.SE - What condensation products would you expect to...Ch. 23.SE - The following reactions are unlikely to provide...Ch. 23.SE - Fill in the missing reagents a-h in the following...Ch. 23.SE - Prob. 70APCh. 23.SE - Prob. 71APCh. 23.SE - Prob. 72APCh. 23.SE - Prob. 73AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- One frequently used method for preparing methyl esters is by reaction of carboxylic acids with diazomethane, CH2N2. The reaction occurs in two steps: (l) protonation of diazomethane by the carboxylic acid to yield methyldiazonium ion, CH3N2+, plus a carboxylate ion; and (2) reaction of the carboxylate ion with CH3N2+. (a) Draw two resonance structures of diazomethane, and account for step 1. (b) What kind of reaction occurs in step 2?arrow_forwardOne step in the urea cycle for ridding the body of ammonia is the conversion of argininosuccinate to the amino acid arginine plus fumarate. Propose a mechanism for the reaction, and show the structure of arginine.arrow_forwardCarboxylic acids having a second carbonyl group two atoms away lose CO2 (decarboxylate) through an intermediate enolate ion when treated with base. Write the mechanism of this decarboxylation reaction using curved arrows to show the electron flow in each step.arrow_forward
- 18-48 4-Aminobenzoic acid is prepared from benzoic acid by the following two steps. Show reagents and experimental conditions to bring about each step.arrow_forwardHeating toluene in the presence of KMnO4 followed by acification with HCl, converts toluene into benzoic acid. Using this information and any other reactions discussed in this course, show a sequence of reactions showing howing toluene can be converted to p-aminobenzoic acid.arrow_forwardPredict the products of the reaction of phenylaceetaldehyde , C6H5CH2CHO, with a. NaBH4 the H3O+ b. Aqueous acidic CrO3 c. NH2OH d.CH3MgBr then H3O+ e, CH3OH,H+ catalystarrow_forward
- Predict the products obtained from the reaction of triolein with the following reagents.(a) NaOH in water (b) H2 and a nickel catalyst (c) Br2 in CCl4(d) ozone, then dimethyl sulfide (e) warm KMnO4 in water (f) CH2I2>Zn(Cu)arrow_forward1,4-Diazabicyclo[2.2.2]octane (abbreviated DABCO) is a tertiary amine that catalyzes transesterification reactions. Explain how it does this.arrow_forwardPropose a structural formula for terpin and a mechanism for its formationarrow_forward
- Show how to synthesize the following amines from the indicated starting materials byacylation–reduction. b) N-benzylaniline from anilinearrow_forwardPredict the chemical name of compound B a. p-chlorobenzylamine b. 4-bromoaniline c. benzylamine d. p-chloronitrile e. p-chlorobenzaldehyde 2. What is the reaction name for the chemical transformation of A to B a. reductive amination b. catalytic reduction c. carbonyl dehydration d. Hofmann elimination e. Aldehyde rearrangementarrow_forwardThe thioester, which form when a Cys side chain condenses with a Gln side chain a few residues away, reacts readily with compounds with the formula ROH or RNH2. Draw the resulting ester and amide reaction products.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License