
(a)
Interpretation:
The other products obtained from the highly stereo and regioselective reaction of O-alkylation during the formation of Gilvarin M. has to be interpreted.
Concept introduction:
Regioselectivity:
Regioselectivity is the preference of one direction of
Stereoselectivity:
Stereoselectivity is the property of a
(a)
Explanation of Solution
The reaction is given as,
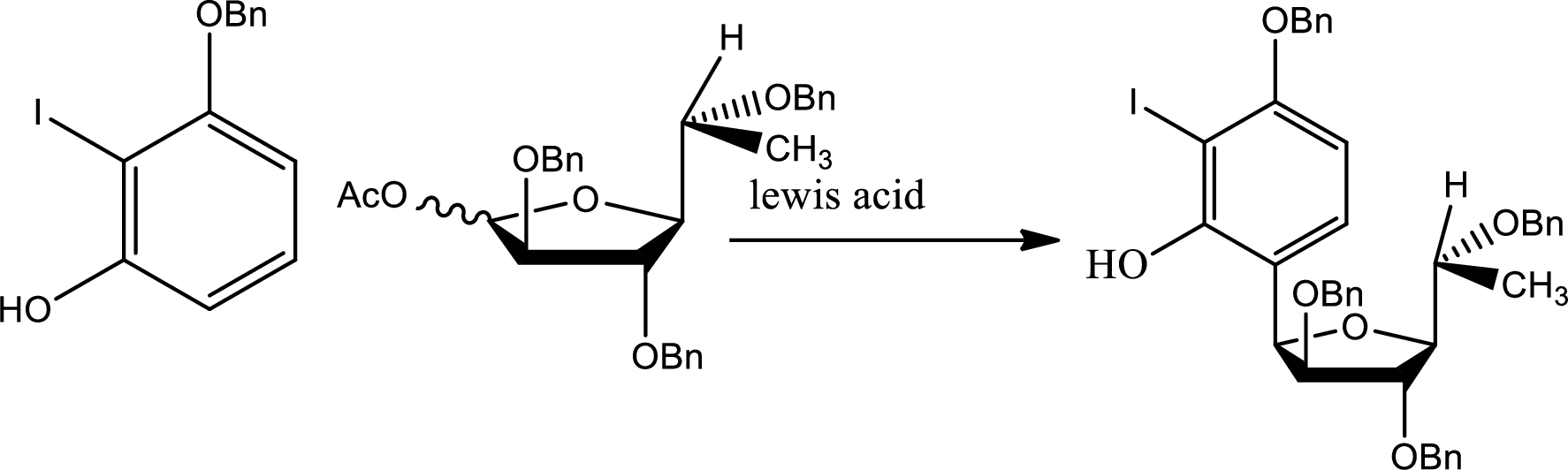
The other possibility is,
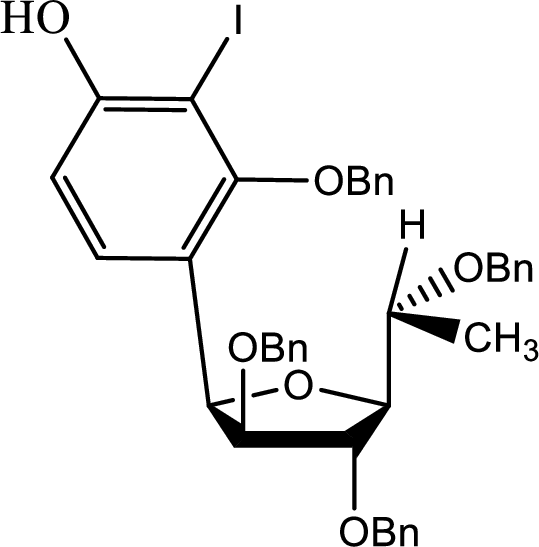
(b)
Interpretation:
The structure of the compound C has to be given.
Concept introduction:
Acid-base reaction:
The species that donates proton or accepts lone pair of electrons are called acids and those who accepts proton or donates lone pair of electrons are called base.
The
(b)
Explanation of Solution
The pathway of formation of C is,
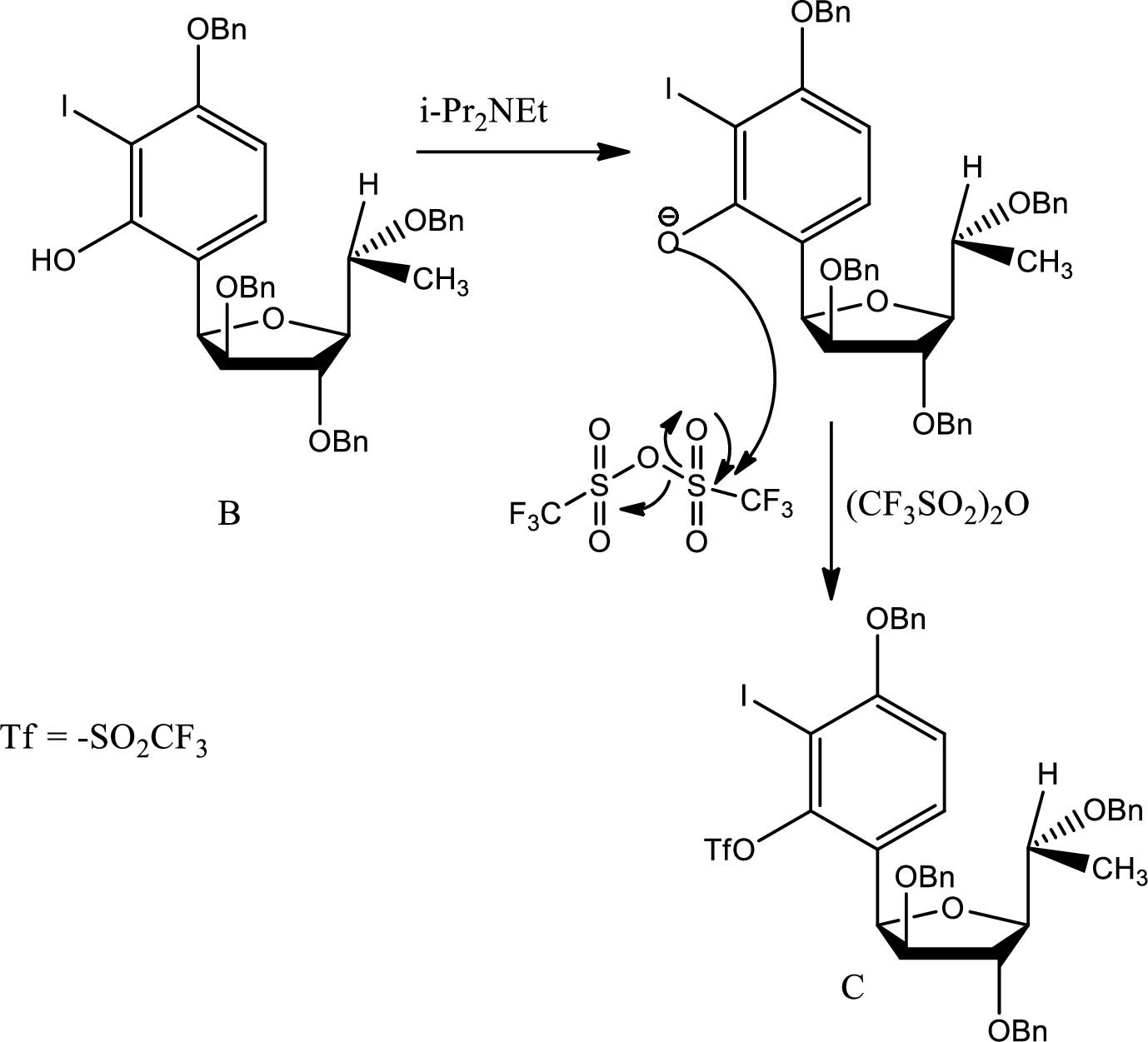
Here the
(c)
Interpretation:
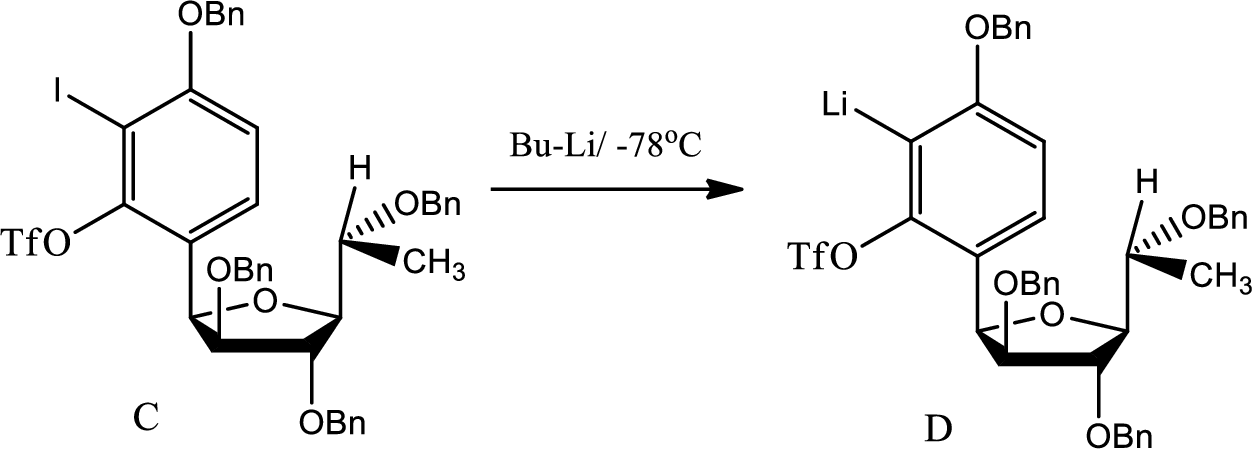
The structure of compound D has to be given.
Concept introduction:
Lithium halogen exchange:
Organolithium reagents are characterised by the presence of
The reaction of lithium metal at low temperature with an
The same reaction happens with the haloarenes also,
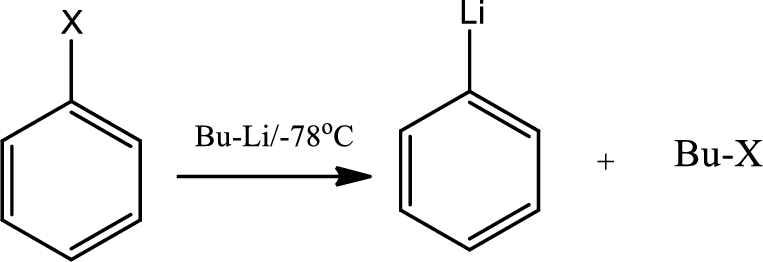
(c)
Explanation of Solution
The reaction for the formation of D is as follows,
(d)
Interpretation:
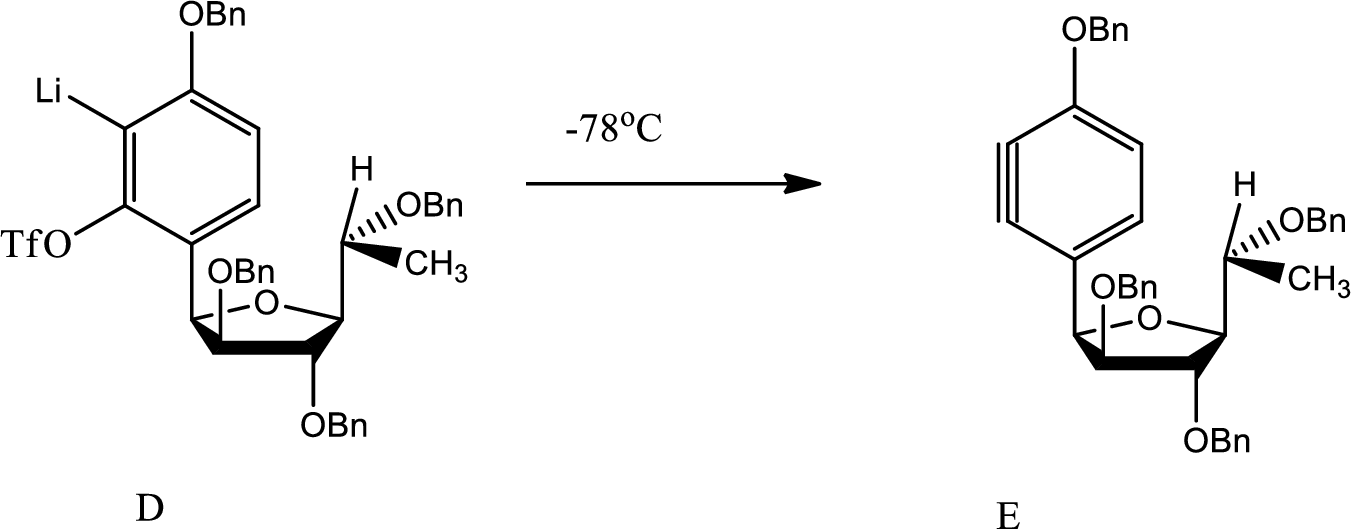
Structure of E has to be given along with the mechanism of formation of E from D.
Concept introduction:
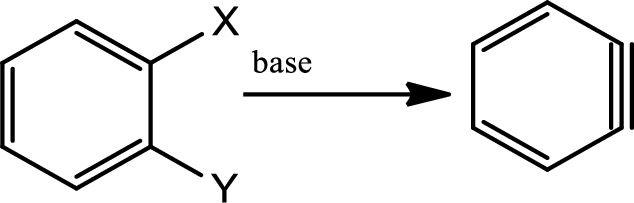
Benzyne formation:
Benzynes are highly reactive intermediate species that are made from
(d)
Explanation of Solution
Here benzyne formation occurs due to removal of
(e)
Interpretation:
A mechanism for formation of F from E has to be given.
Concept introduction:
Diels Alder reaction:
The Diels-Alder reaction is a chemical reaction between a conjugated diene and a substituted
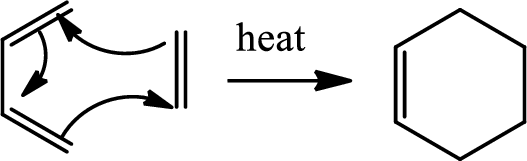
(e)
Explanation of Solution
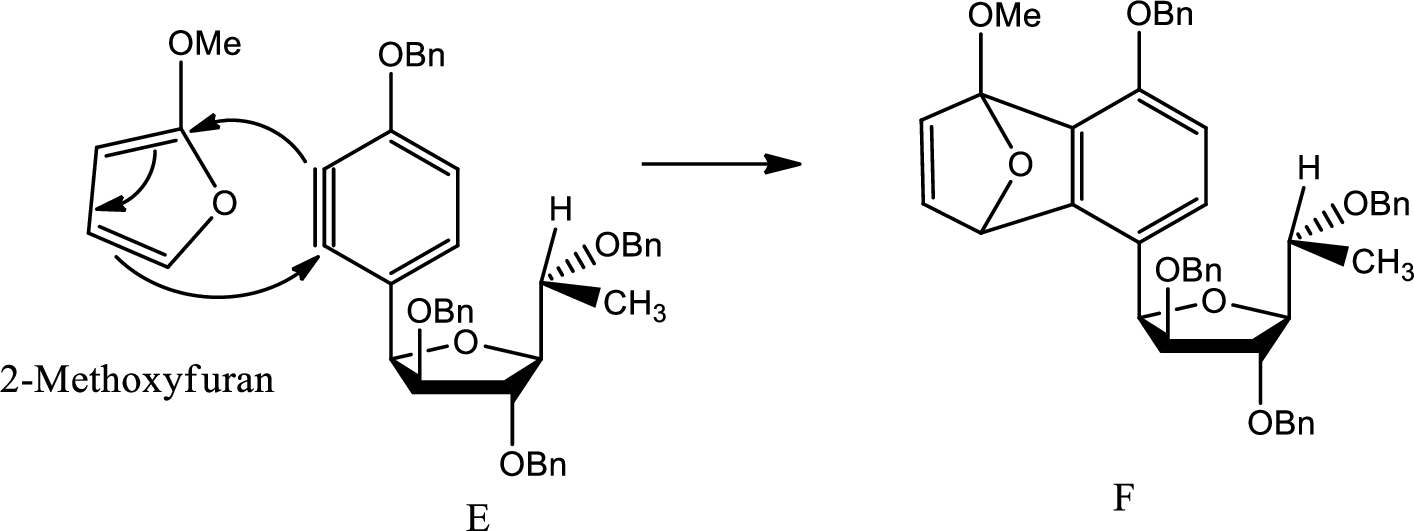
Here Diels Alder reaction occurs between furan and benzyne. A new ring is formed.
(f)
Interpretation:
Mechanism from F to G has to be given.
Concept introduction:
Hydrolysis:
Hydrolysis is any chemical reaction in which a water molecule ruptures one or more chemical bonds. This is mainly used for substitution, elimination and fragmentation reactions in which water is the nucleophile. Hydrolysis is reverse of condensation reaction because in this process water is added to beak down.
(f)
Explanation of Solution
Here aqueous work up leads to breaking of the bridging oxygen bond by protonating the oxygen. Thus the angle strain increases as positive charge on electronegative oxygen is tough. Thus the bond breaks and the charge formed is stabilised by electron donating effect of
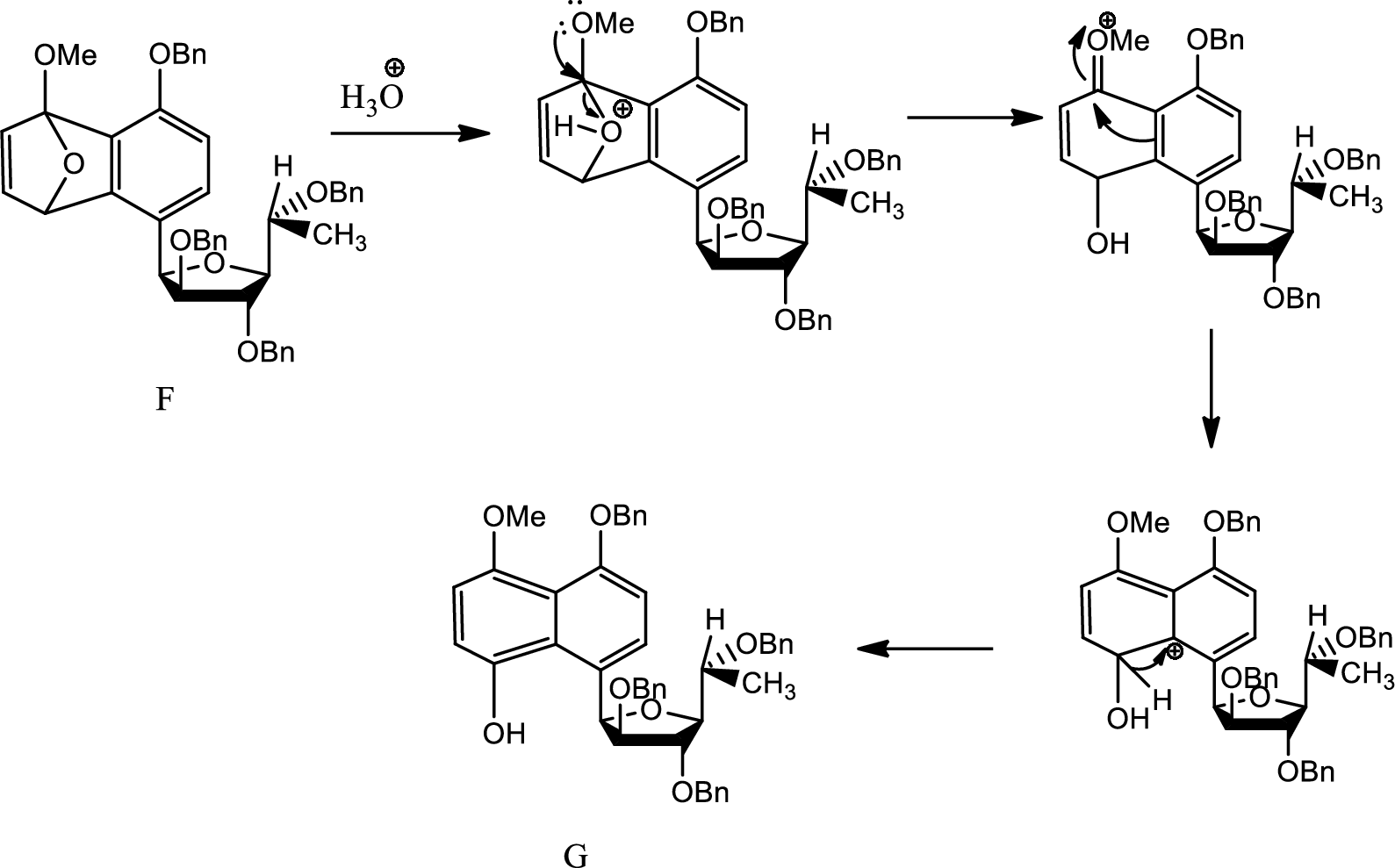
Thus G is formed from F.
(g)
Interpretation:
The reagents and condition from G to H has to be given.
Concept introduction:
Acid-base reaction:
The species that donates proton or accepts lone pair of electrons are called acids and those who accepts proton or donates lone pair of electrons are called base.
The
(g)
Explanation of Solution
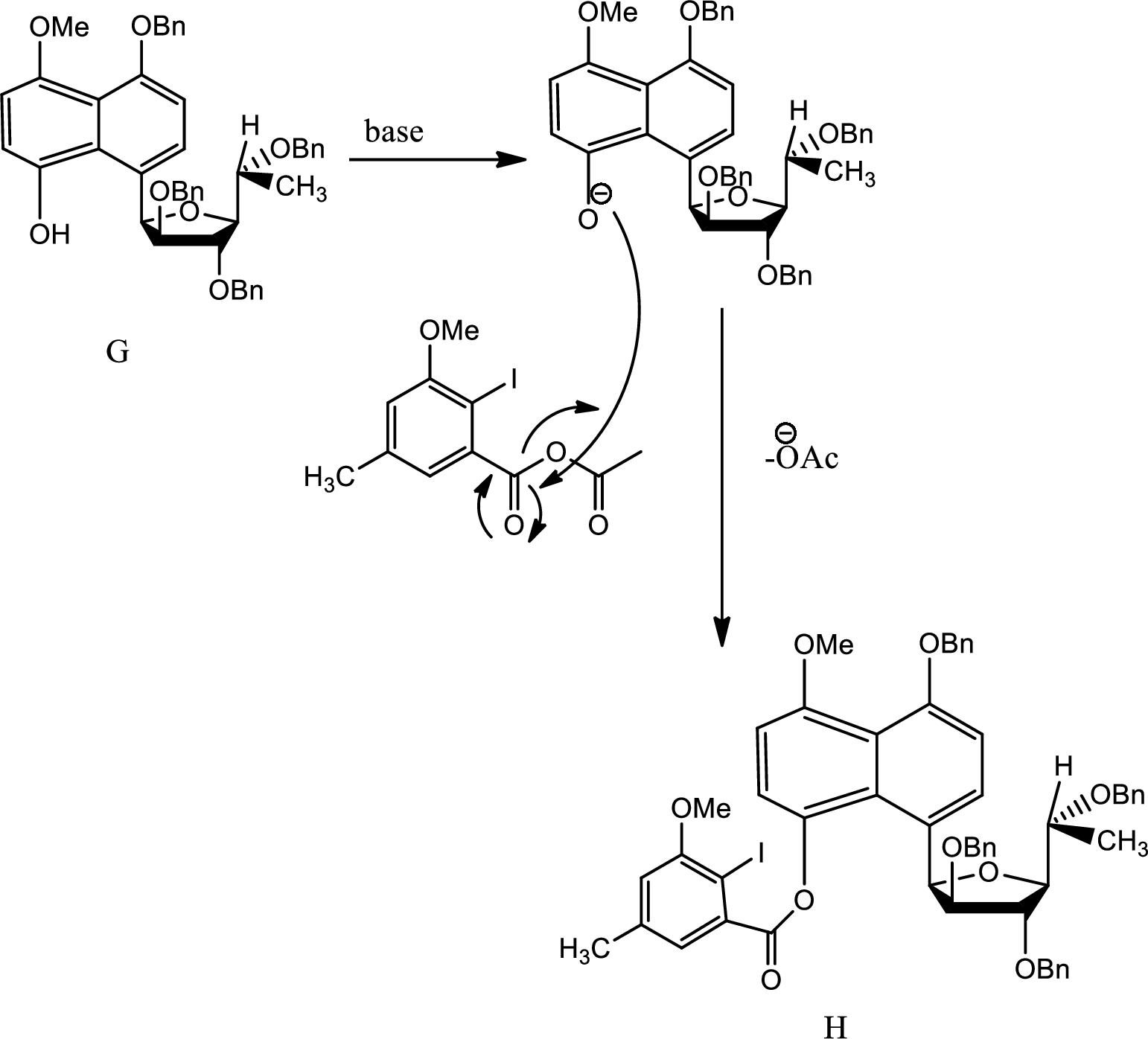
Here on giving base the acidic hydrogen from phenol is removed and the new carbanion formed undergoes
Thus H is formed from G.
(h)
Interpretation:
Reagents and condition for conversion of H to I has to be given.
Concept introduction:
Cross coupling:
A cross coupling reaction is defined as a reaction that creates a
In the case of palladium catalysed cross-coupling reactions the other metal or metalloids are commonly
Suzuki coupling:
The Suzuki coupling uses a boron compound and an alkenyl, aryl or alkyl halide or triflate as the carbon sources with a palladium salt as a catalyst. The reaction is mainly used to form biaryls. The mechanism of the reaction starts with an oxidative addition followed by transmetallation in which the substituent on the borane replaces the ligand on the palladium concluding with the reductive elimination of the palladium to form the new carbon-carbon bond. The base may serve as a new labile ligand to palladium or it may activate the borane by coordination.
Generalized reaction,
Oxidative addition and ligand exchange,
Borane activation
Reaction,
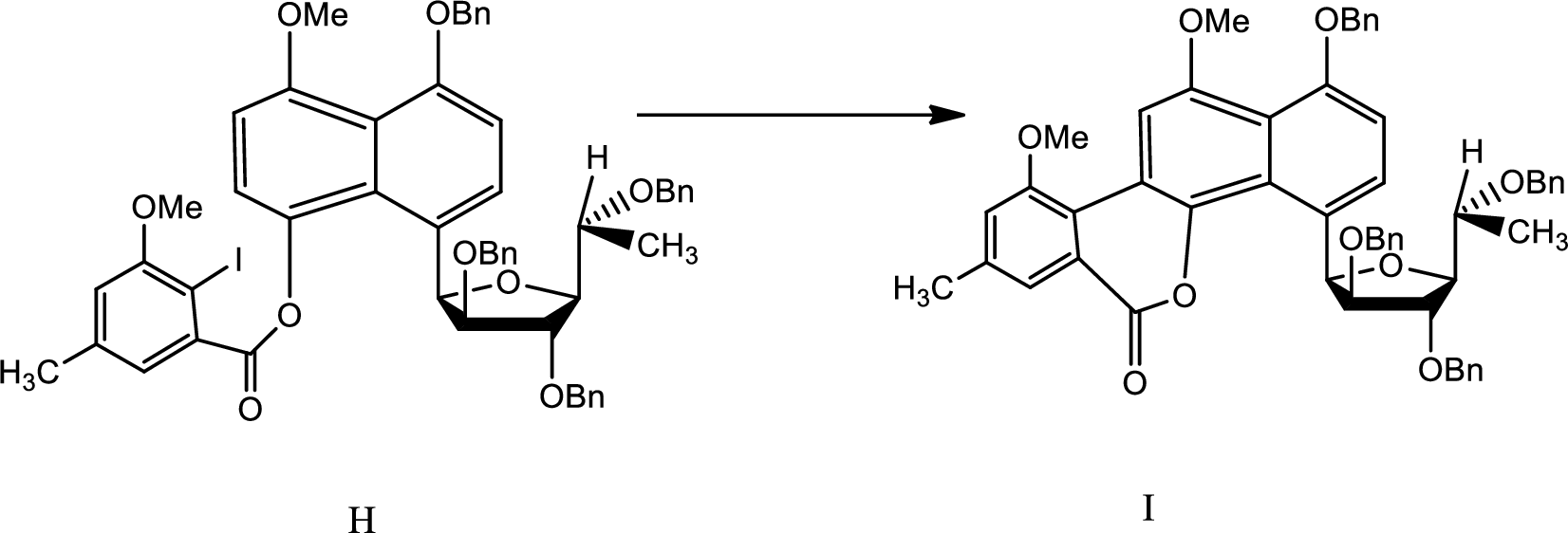
(h)
Explanation of Solution
The reaction is given as,
The reaction proceeds as follows,
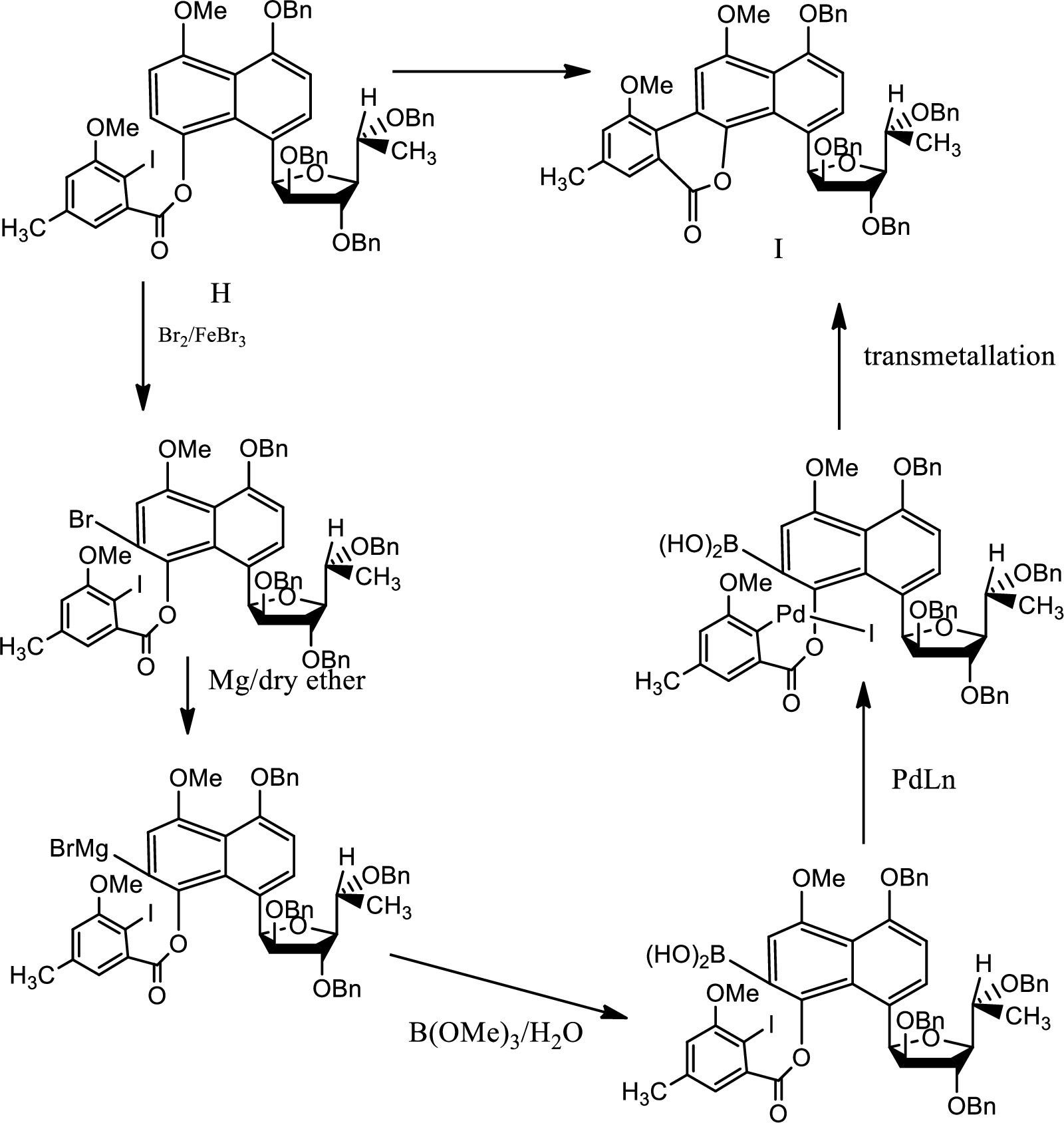
1st boron has to be added so that via transmetallation by intramolecular Suzuki coupling the product can be formed.
(i)
Interpretation:
Reagent needed to form Gilvocarcin M. from I has to be interpreted.
Concept introduction:
Protection-deprotection:
A protecting group is introduced to a molecule by chemical modification of a
Hydrogenolysis:
Hydrogenolysis is a chemical reaction whereby a carbon carbon bond or carbon heteroatom single bond is cleaved by hydrogen gas catalytically.
(i)
Explanation of Solution
The reaction is given as,
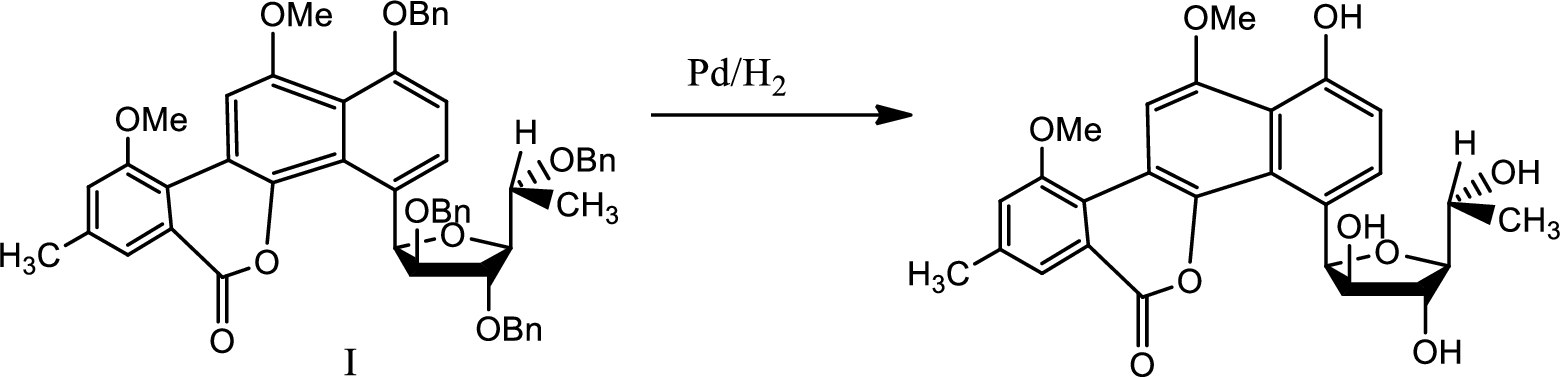
Here deprotection is done by hydrogenolysis with the help of palladium and catalytic amount of hydrogen.
(j)
Interpretation:
Probable source of chiral centres has to be found.
Concept introduction:
Chiral centre:
Chiral centre is defined as an atom bonded to four different chemical species. It is a stereo centre that holds the atom in such way that the structure may not be superimposable to its mirror image. They give optical isomerism.
(j)
Explanation of Solution
The source of the chiral centre is only the five membered carbohydrate ring attached to the benzene ring as shown below,
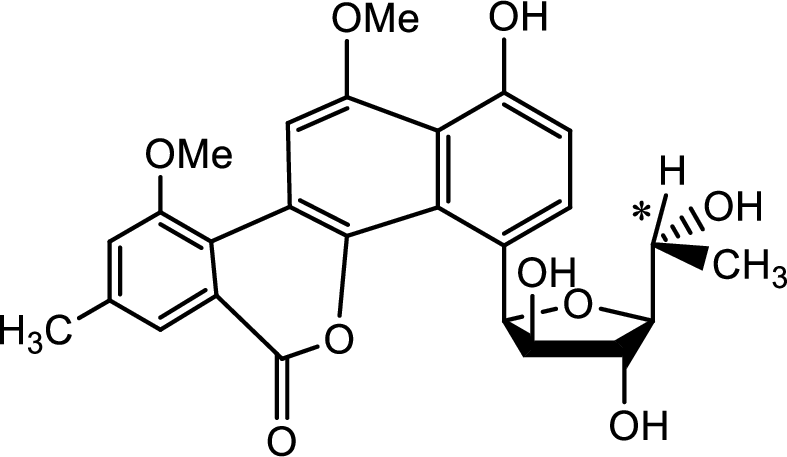
The chiral centre is marked with the asterisk.
(k)
Interpretation:
The need of protection of hydroxyl group has to be stated.
Concept introduction:
Protection-deprotection:
A protecting group is introduced to a molecule by chemical modification of a functional group to obtain chemoselectivity in a subsequent chemical reaction specially in multistep organic synthesis.
(k)
Explanation of Solution
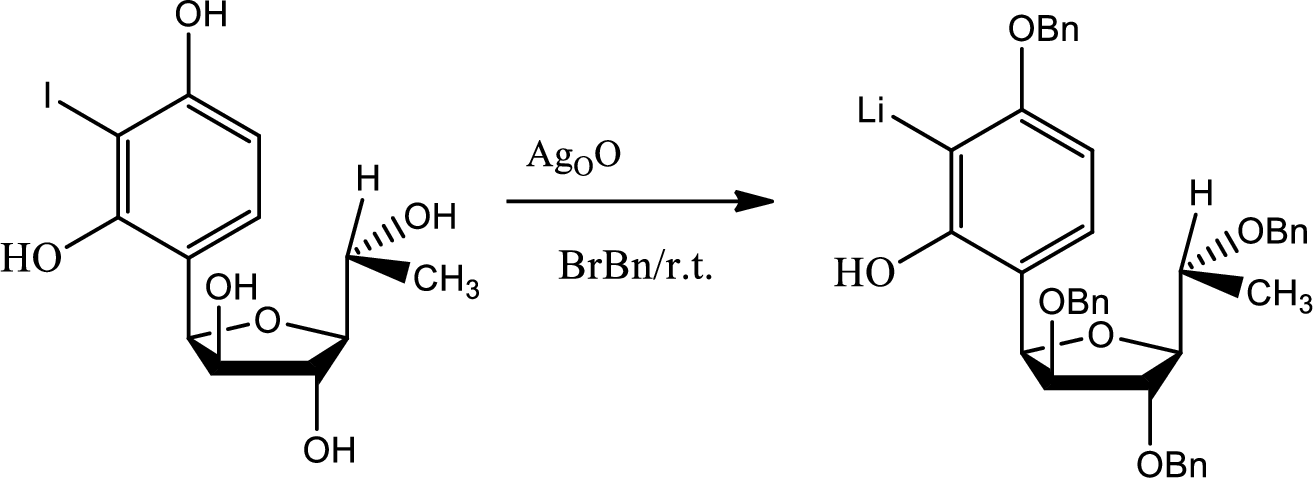
If protection has not been done to the hydroxyl groups then there were possibilities of getting so many unwanted products.
During the acid base reaction all the hydroxyl groups could have lost the proton and thus all the substitution could have taken place in different places. The benzyne formation could have hampered and the stereochemistry could have changed.
The process of protection to the hydroxyl group is given by,
Want to see more full solutions like this?
Chapter 24 Solutions
ORGANIC CHEMISTRY-OWL V2 ACCESS
- Following is the structure of miconazole, the active antifungal agent in a number of over-the-counter preparations, including Monistat, that are used to treat vaginal yeast infections. One of the compounds needed for the synthesis of miconazole is the trichloro derivative of toluene shown on its right. (a) Show how this derivative can be synthesized from toluene. (b) How many stereoisomers are possible for miconazole?arrow_forward7) Explain why the diastereoselectivity is so much better in the first reaction versus the second one. Ph- Ph- m-СРВА + errintioner 95:5 anti:syn Ph- Ph- m-CPBA + enantiomer 61:39 canti:synarrow_forwardIdentify the FALSE statement pertaining to SN1 and SN2 mechanisms: An SN2 reaction will result in an inversion of configuration (stereochemistry). An SN1 substitution will result in inversion and retention of configuration (stereochemistry). O in In the reaction of OH with CICH₂CH₂CH3, there is no carbocation intermediate. An SN1 substitution occurs in more than 1 step. In the reaction of H₂O with CIC(CH3)3, increasing the concentration of CIC(CH3)3 will not increase the rate of reaction.arrow_forward
- 6) Give the structures of the compounds A-E in the synthetic sequences below. PBr3, Br2 NH3, excess LOH A B you 1) NaNH2 (1 equiv) H2O, HgSO4 Br2 excess HCECH C D E H2SO4 NaOH excess 2) BuCI (1 equiv)arrow_forwardPropose a plausible mechanism for the following transformation and justify the stereochemical outcome (J. Org. Chem. 1999, 64. 4617-4626): CO₂Me Heat MeOH MeO ° CO₂Me Step 1 Add any remaining curved arrow(s) to draw the first step of the mechanism. H₁₂C H.C ་་་་ H₂C O-CH3 Edit Drawing CH₂arrow_forwardWhich reagent(s) would be the best choice for completing the sequence shown? OH NaBH4, MeOH MeOH [TSOH] Dean-Stark Trap O 1) LiAlH4, 2) H₂O O 1) DIBAL-H, -78°C 2) H₂O O LIAI(OtBu)3H Harrow_forward
- Which of the following reactions describes why AICI3 needs to stay dry in order for it to be used in a Freidel Crafts Acylation? A) 2 AIC13 + 6H2O → 2 Al(OH)3 (s) + 3 Cl2(g) + 6H2(g) B) AIC13 + 6H2O → [[Al(H2O)5 (OH)]+ H+ + 3 CI C) AlCl3 +H2O → HAICI3* + OH¯ D) All the reactions, listed above, are competing for the Alš+ ionarrow_forwardAcid anhydrides are often used in place of acid chlorides because a less acidic carboxylic acid, not the much stronger acid HCI, is the byproduct of the reaction. In the following reaction of a carbohydrate derivative, acetic anhydride is used to obtain the product in 99% yield as a single stereoisomer. Note that the stereochemistry of the starting ano- meric carbon is not indicated. Draw the product of the following transformation in a chair form and show the single stereoisomer product of this transformation, which is also the most stable possible chair species. OH anomeric carbon H3C "O-CH3 CH,Cl2 Siarrow_forwardSodium ethoxide is formed when ethanol (pKa 15.8) is mixed with sodium metal, .Which among the compounds below will favorably react with sodium ethoxide through an acid/base reaction? CH3CO2H (pKa 4.8), H-C≡C-H (pKa 25), H2O (pKa 14.0), NH3 (pKa 38) Pls draw the mechanism for the favored reaction.arrow_forward
- (i) (ii) Identify the alpha protons in ethyl acetoacetate (shown below) and draw the structures of the two different enolates which would form under basic conditions. Explain why one enolate is more stable than the other. L ملو Ethyl acetoacetate Draw the structure of the product N formed in the crossed aldol reaction between ketone L and aldehyde M (no mechanism required). H OEt M NO₂ NaOH H₂O/EtOH N C15H14NO4arrow_forwardLike alcohols, ethers undergo α cleavage by breaking a carbon–carbon bond between an alkyl group and the carbon bonded to the ether oxygen atom; that is, the red C–C bond in R–CH2OR' is broken. With this in mind, propose structures for the fragments formed by α cleavage of (CH3)2CHCH2OCH2CH3. Suggest a reason why an ether fragments by α cleavage.arrow_forwardDraw the structures of compounds A and B in the scheme below. Be sure to indicate the stereochemistry. E2 Ph Elarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


