
Concept explainers
(a)
Interpretation:
The number of acetyl CoA is formed from complete beta-oxidation of the fatty acid depicted in the ball and stick model, needs to be identified.
Concept Introduction:
Beta − oxidation of fatty acids involves four series of reactions. In this reaction, the long-chain fatty acid is degraded into many two-carbon unit's acetyl CoA.
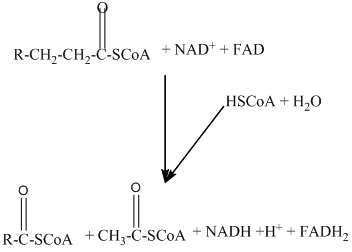
The number of acetyl CoA molecules formed, and the number of the time beta-oxidation occurs can be determined by using the number of carbon atoms present in the fatty acid.
(b)
Interpretation:
The number of cycles of beta-oxidation is required for complete oxidation needs to be identified.
Concept Introduction:
Beta − oxidation of fatty acids involves four series of reactions. In this reaction, the long-chain fatty acid is degraded into many two-carbon unit's acetyl CoA.
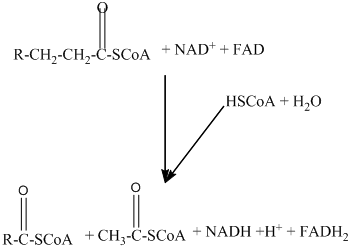
The number of acetyl CoA molecules formed, and the number of time beta-oxidation that occur can be determined by using the number of carbon atoms present in the fatty acid.
Want to see the full answer?
Check out a sample textbook solution
Chapter 24 Solutions
CONNECT IA GENERAL ORGANIC&BIO CHEMISTRY
- Fatty acids and triglycerides are an important source of nutrition and a dense form of stored energy. Digestion of fats yields more energy per gram than digestion of carbohydrates. Each turn on the B-oxidation spiral results in the formation of a new acetyl COA molecule. This molecule can then be used to generate more energy in A) fatty acid synthesis B) ketolysis C) glycolysis D) tricarboxylic acid cycle E) oxidative phosphorylationarrow_forwardWhich of the following are triacylglycerols? a) sphingomyelins; e) fats; b) oils; f) sphyngolipids; c) steroids; g) prostaglandins; d) terpenes; h) waxes.arrow_forwardIf an animal has 5.000 kg of palmitoyl-CoA available for fatty acid oxidation, how many kilograms of water could the animal produce from that palmitoyl-CoA? The chemical formula for palmitoyl-CoA is C37H66N7O17P3S.arrow_forward
- Which of the following are saponifiable lipids? a) triacylglycerols; f) stepoids; b) prostaglandins; g) glycolipids; c) waxes; h) terpenes; d) leukotrienes; i) phosphoglycerides; e) sphyngolipids; k) plasmalogens? Write the equation showing triacylglycerol formation from 1 mol of glycerol and 1 mol each of oleic, stearic and palmitic acids. Write the equation for the acid-catalyzed hydrolysis of the triacylglycerol: Write the equation for the saponification of the triacylglycerol: Write the equation for the hydrogenation of the triacylglycerol: Write the structure of the phosphoglyceride formed from 1 mol each of glycerol, stearic and palmitic acids, phosphoric acid, and choline . Show the formation of the sphingolipid containing oleic acid, phosphoric acid,…arrow_forwardStearic acid is the common name for the C18 straight-chain, saturated fatty acid. Draw the structure of the fatty acyl-CoA that this forms (you don't have to draw out all of the atoms in coenzyme A; you can represent it as S-, and then show all of the reactions). Draw & identify all of the compounds formed when the eighteen carbon fat is converted to two molecules of acetyl-CoA and the fourteen-carbon fat via two turns of the fatty acid spiral. List the enzymes and coenzymes that are part of the pathway.arrow_forwardWhat is the product of the reaction when α-amylase acts on amylose? (A) amylose molecules (B) monosaccharide molecules (C) different-sized oligosaccharide fragments (D) different-sized polysaccharide fragmentsarrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning



