
The crystal field stabilization energy (CFSE) can be defined as CFSE = 31 number of electrons the dxy, dxx, and dyzorbitals are stabilized (lower energy) with respect to the average energy of thed orbitals and that the deand dr, orbitals are destabilized. As described in Are You Wondering 24-3, the stabilization is -0.4
a. Plot the hydration energies as a function of the
b. Assuming that all the hexaaqua complexes are high spin, which ions have zero CFSE?
c. If lines are drawn between those ions with CFSE = 0, a line of negative slope is obtained. Can you explain this finding?
d. The ions that do not have CFSE = 0 have heats of hydration that are more negative than the lines drawn in part (c). What is the explanation for this?
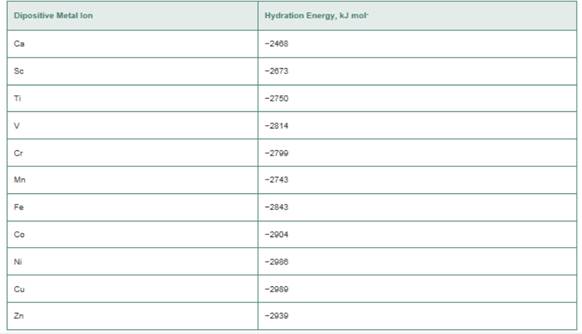
e. Estimate the value of L. for the Fe(ll) ion in an octahedral field of water molecules. What
Want to see the full answer?
Check out a sample textbook solution
Chapter 24 Solutions
GENERAL CHEMISTRY(LL)-W/MASTERINGCHEM.
- Give the coordination numbers and write the formulas for each of the following, including all isomers where appropriate: (a) tetrahydroxozincate(ll) ion (tetrahedral). (b) hexacyanopalladate(IV) ion. (c) dichlomaurate(I) ion (note that aurum is Latin for "gold"). (d) diamminedichloroplatinum(Il). (e) potassium diamminetetrachlorochromate(III). (f) hexaamminecobalt(III) hexacyanochromate(III). (g) dibromobis(ethylenediamine) cobalt(III) nitratearrow_forwarda. In the absorption spectrum of the complex ion Cr(NCS)63, there is a band corresponding to the absorption of a photon of light with an energy of 1.75 104 cm-1. Given 1 cm1 = 1.986 1023 J, what is the wavelength of this photon? b. The CrNC bond angle in Cr(NCS)63 is predicted to be 180. What is the hybridization of the N atom in the Ncs- ligand when a Lewis acid-base reaction occurs between Cr3+ and NCs- that would give a 180 CrNC bond angle? Cr(NCS)63 undergoes substitution by ethylenediamine (en) according to the equation Cr(NCS)63+2enCr(NCS)2(en)2++4NCS Does Cr(NCS)2(en)2+ exhibit geometric isomerism? Does Cr(NCS)2(en)2+ exhibit optical isomerism?arrow_forwardGive the coordination number for each metal ion in the following compounds: (a) [Co(CO3)3]3- (note that CO32- is bidentate in this complex). (b) [Cu(NH3)4]2+. (c) [Co(NH3)4Br2]2(SO4)3. (d) [Pt(NH3)4][PtCl4]. (e) [Cr(en)3](NO3)3. (f) [Pd(NH3)2Br2] (square planar). (g) K3[Cu(Cl)5]. (h) [Zn(NH3)2Cl2]arrow_forward
- The factor determining the mechanism observed in the substitution reactions of square plane complexes is that these complexes are not saturated with coordination. Right?False?arrow_forwardThe values for lmax for complex A and B are 512 nm and 650 nm respectively. A has the structure [Co(NH3)4CO3]NO3 while B is [Co(NH3)4Cl2]Cl. Explain this data based on the Crystal Field Theory UV-visible spectroscopy provides information about cobalt(III) complexes. A blue cobalt complex has molar absorbtivity of 125 Lmol-1cm-1. Is the complex octahedral or tetrahedral? Justify your answer and explain the colour of the complex.arrow_forwardMn(acac)3 was found to have its ground state to 1st excited state absorption at 500 nm (Δo). The pairing energy for this complex is 28,000 cm-1 . Based on this information, draw the populated ground state d orbital diagram for this complex.arrow_forward
- Predict the geometry and number of unpaired spins inthe following four-coordinate complexes: [Ni(CN)4]2-,[NiCl4]2-. Explain your reasoning.arrow_forwardThe periodic trend which is responsible for the observation that the maximum oxidation state of the transition-metal elements peaks near groups 7B and 8B?arrow_forwardThe mineral magnetite has the formula Fe3O4 and has an inverse spinel structure. In the unit cell, there are two Fe ions in octahedral sites and one Fe ion in a tetrahedral site. (i) Are the Fe ions in magnetite low-spin or high-spin? Explain your reasoning. (ii) Draw d-orbital splitting diagrams for Fe(III) in tetrahedral and octahedral ligand fields and hence deduce the CFSE in each case. (iii) On the basis of your answer to part (ii) above, do you expect Fe(III) to have a preference for tetrahedral or octahedral sites? Explain your answer. (iv) Draw d-orbital splitting diagrams for Fe(II) in tetrahedral and octahedral ligand fields and hence deduce the CFSE in each case. (v) Assuming that Δt ~ 4/9 Δo and using your answer to part (iv) above, deduce the CFSE of Fe(II) in a tetrahedral field in terms of Δo. Hence decide whether the tetrahedral site is occupied by Fe(II) or Fe(III), explaining your reasoning.arrow_forward
- Give a scientific reason for each of the following: 1- No known tetrahedral ionic complexes.(Pd + 2, Pt + 2 ) 2- The strength of the ligands CN, CO ? 3- The cleavage of d orbitals in C.F.T. by the influence of the ligands, where the high level eg and the low level of t2g are observed. 4- Werner's coordination theory succeeded in explaining its complexes. Subject: inorganic chemistry I hope for a quick solutionarrow_forwardThe maximum absorption for the complex ion [Co(NH3 )6 ] 3+ is at 470 nm. A) Predict the color of the complex and B) calculate the division of the crystalline field in kJ/mol and cm-1arrow_forwardAt 580 nm, the wavelength of maximum absorption, the complex Fe(SCN)2+ has a molar absorptivity of 7.00 x 103 M-1 cm-1. Calculate, (a) the absorbance of a 3.50 x 10-5 M solution of the complex at 580 nm in a 1.50 cm cell (b) the absorbance of a solution in a 2.00 cm cell in which the concentration of the complex is half that in (a)arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





