
Interpretation:
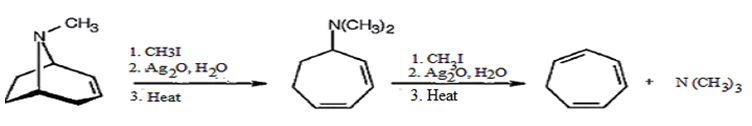
How to convert tropidene in to tropilidene (1,3,5-cycloheptatriene) is to be shown.
Concept introduction:
Two successive Hofmann elimination reactions of with quarternary ammonium salt of tropidene will yield tropilidene.
To show:
How to convert tropidene in to tropilidene (1,3,5-cycloheptatriene).
Answer:
The conversion of tropidene in to tropilidene (1,3,5-cycloheptatriene) is given below.
Explanation:
Tropidene when treated with excess of methyl iodide yields a quartenary ammonium salt which undergoes Hofmann elimination when heated with Ag2O and water to produce N,N-dimethylcycloheptadiene. Methylation of the diene with excess of methyl iodide yields another quartenary ammonium salt which undergoes Hofmann elimination reaction to yield tropilidene.
Conclusion:
The conversion of tropidene in to tropilidene (1,3,5-cycloheptatriene) is given below.

Want to see the full answer?
Check out a sample textbook solution
Chapter 24 Solutions
Organic Chemistry
- Treatment of a cyclic ketone with diazomethane is a method for accomplishing a ring-expansion reaction. For example, treatment of cyclohexanone with diazomethane yields cycloheptanone. Propose a mechanism.arrow_forwardShow the products from reaction of p-bromoaniline with the following reagents: (a) CH3I (excess) (b) HCl (c) HNO2, H2SO4 (d) CH3COCl (e) CH3MgBr (f) CH3CH2Cl, AlCl3 (g) Product of (c) with CuCl, HCl (h) Product of (d) with CH3CH2Cl, AlCl3arrow_forwardIn Problem 7.28, we saw this two-step sequence in Johnsons synthesis of the steroid hormone progesterone. Propose a structural formula for the intermediate formed in Step 3 and a mechanism for its conversion in Step 4 to progesterone.arrow_forward
- The Favorskii reaction involves treatment of an -bromo ketone with base to yield a ring-contracted product. For example, reaction of 2-bromocyclohexanone with aqueous NaOH yields cyclopentanecarboxylic acid. Propose a mechanism.arrow_forwardShow how m-toluidine can be converted tom-toluidineCH3 NH2the following compounds, using any necessary reagents.m-toluonitrile(a) CH3 C N CH3 CH2NH2m-methylbenzylamine(b)m-iodotoluenearrow_forwardWhat product is formed when 2,2-dimethyloxirane is treated with each set ofreagents: −OCH3 followed by H2O, or CH3OH and H2SO4?arrow_forward
- What is the NAME of the substrate product when benzene is first treated with HNO3 and H2SO4, and THEN treated (in a second step) with Br2, and FeCl3 (or FeBr3)?arrow_forwardShow how m-toluidine can be converted tom-toluidineCH3 NH2the following compounds, using any necessary reagents.m-toluonitrilearrow_forwardWhich one the following compounds on treatment with Grignard reagent followed by hydrolysis produce CH3CH2COOH? CH3CHO CH3COCH3 CO2 HCHOarrow_forward
- Mustard gas, Cl¬CH2CH2¬S¬CH2CH2¬Cl, was used as a poisonous chemical agentin World War I. Mustard gas is much more toxic than a typical primary alkyl chloride. Itstoxicity stems from its ability to alkylate amino groups on important metabolic enzymes,rendering the enzymes inactive.(a) Propose a mechanism to explain why mustard gas is an exceptionally potent alkylatingagentarrow_forwardUse the following choices for questions 36-40: SN1 SN2 E1 E2 E1cb 36. A polar aprotic solvent is preferred in this reaction. 37. Product has an inverted stereochemistry. 38-39. Proceeds in a concerted path. 40. A mixture of products is obtained in this reaction.arrow_forwardThank you for the answer, would it be possible to follow the path I have attached as well srarting from methanol ---> CH3Br -----> CH3MgBr-------> (H3C)CH-CH2OH ----> E1 Isobutalenearrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

