
Concept explainers
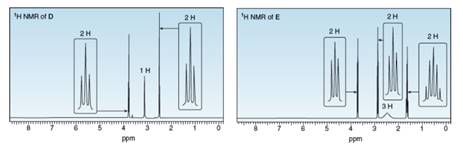
Treatment of compound D with LiAlH4 followed by H2O forms compound E. D shows a molecular ion in its mass spectrum at
Trending nowThis is a popular solution!

Chapter 25 Solutions
ORGANIC CHEMISTRY-W/ACCESS
- C9H10O2: IR absorption at 1718 cm–1. Propose a structure with data given.arrow_forwardTreatment of anisole (CH3OC6H5) with Cl2 and FeCl3 forms P, which has peaks in its mass spectrum at m/z = 142 (M), 144 (M + 2), 129, and 127. P has absorptions in its IR spectrum at 3096–2837 (several peaks), 1582, and 1494 cm-1. Propose possible structures for P.arrow_forwardcompound with the molecular formula C7H9N exhibits IR bands at 3450 cm-1 (medium, doublet), and 855 cm-1 (strong) and shows the following major mass spectral signals (m/z): 106 (base); 107 (M+, about 70% of base), 91 (40% of base), and 77 (about 20% of base). Deduce a reasonable structure from this data.arrow_forward
- Treatment of benzoic acid (C6H5CO2H) with NaOH followed by 1-iodo-3-methylbutane forms H. H has a molecular ion at 192 and IR absorptions at 3064, 3035, 2960–2872, and 1721 cm-1. Propose a structure for H.arrow_forwardDraw the structure of a compound with the formula C5H10O2 (along with the reasons of choosing it) which, upon analysis, gave key peaks in an infrared spectrum at 3450 cm-1 and 1713 cm-1, as well as the following 1H-NMR spectrum.arrow_forwardAn organic compound B with formula C6H14O has the following: IR Spectroscopy 2974 cm-1, 1080 cm-1 Mass Spectrometry 102 (M+), 87, 73 1H NMR Spectroscopy Eight signals at δ 1.10 (d, 3H), 1.13 (dd, 3H), 1.14 (dd, 3H), 1.59 (ddq, 1H), 1.60 (ddq, 1H), 3.19 (ddq, 1H), 3.51 (dq, 1H), 3.50 (dq, 1H). Compound B is obtained by the reaction of compound A with NaH followed by CH3CH2Br. The stereochemistry of A is "S" Using this information, deduce a plausible structure for Compound A with correct stereochemistry.arrow_forward
- Deduce the structures of compounds A and B, two of the major components of jasmine oil, from the given data. Compound A: C9H10O2; IR absorptions at 3091–2895 and 1743 cm-1; 1H NMR signals at 2.06 (singlet, 3 H), 5.08 (singlet, 2 H), and 7.33 (broad singlet, 5 H) ppm. Compound B: C14H12O2; IR absorptions at 3091–2953 and 1718 cm-1; 1H NMR signals at 5.35 (singlet, 2 H) and 7.26–8.15 (multiplets, 10 H) ppm.arrow_forwardFollowing are IR and 1H-NMR spectra of compound D. The mass spectrum of compound D shows a molecular ion peak at m/z 136, a base peak at m/z 107, and other prominent peaks at m/z 118 and 59. Q.) Propose structural formulas for ions in the mass spectrum at m/z 118, 107, and 59.arrow_forwardCompounds B and C are isomers with molecular formula C5H9BrO2. The 1H NMR spectrum of compounds B and C are shown below. The IR spectrum corresponding to compound B showed strong absorption bands at 1739, 1225, and 1158 cm-1, while the spectrum corresponding to compound C have strong bands at 1735, 1237, and 1182 cm-1. 1.Based on the information provided, determine the structure of compounds B and C. 2.Assign all peaks in 1H NMR spectrum of compounds B and C.arrow_forward
- Deduce a possible structure for the compound with the IR absorptions below. C5H8O: 2950, 1750 cm-1 C4H8O: 2950, 2820, 2715, 1715 cm-1arrow_forward3-Chlorocyclopropene, on treatment with AgBF4, gives a precipitate of AgCl and a stable solution of a product that shows a single 1H NMR absorption at 11.04 δ. What is a likely structure for the products, and what is its relation to HĂ¼ckel’s rule?arrow_forwardAddition of m-xylene to the strongly acidic solvent HF/SbF5 at 45C gives a new species, which shows 1H-NMR resonances at 2.88 (3H), 3.00 (3H), 4.67 (2H), 7.93 (1H), 7.83 (1H), and 8.68 (1H). Assign a structure to the species giving this spectrum.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

