
Concept explainers
(a)
Interpretation:
The role of the process of diffusion in the transport of gases through the body needs to be explained.
Concept Introduction:
The gasses are transported from the lungs to the whole body and this transfer of gases in the lungs takes place by the process of diffusion. The gases in the lungs diffuse from their region of high pressure to their region of low pressure and are either taken throughout the body or exhale out depending upon the gas.
(b)
Interpretation:
The role of carbaminohemoglobin in the transport of gases throughout the body needs to be explained.
Concept Introduction:
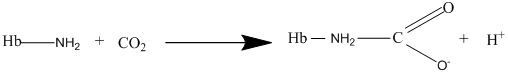
Other than diffusion, there are other methods for the transport of gases. The carbaminohemoglobin is one of the methods of transport for the carbon dioxide gas where the carbon dioxide binds with hemoglobin to form carbaminohemoglobin as per the following equation:
(c)
Interpretation:
The role of alveoli in the transfer of gases throughout the body needs to be explained.
Concept Introduction:
Alveoli are the air sacs in the lungs which basically increases the surface area capacity of the lungs and these alveoli are the sites of the gases transfer from the atmosphere to the blood in the body.
(d)
Interpretation:
The role of carbonic anhydrase in the transfer of gases throughout the body needs to be explained.
Concept Introduction:
The transfer of carbon dioxide also takes place by means of carbonic anhydrase. The carbonic anhydrase converts to carbonic acid

Want to see the full answer?
Check out a sample textbook solution
Chapter 25 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- The process of lysing occurs when the concentration outside a cell is and is said to be lower in concentration; hypotonic higher in concentration; hypertonic higher in concentration; hypotonic lower in concentration; hypertonicarrow_forwardWhat is Osmatic Pressure?arrow_forwardWhich of the following can pass through a dialysis membrane? Ocolloidal particles O macromolecules small molecules all of themarrow_forward
- What is the volume (in liters) occupied by 49.8 g of HCI at STP?arrow_forwardA 70kg man expends 480 kcal of energy per hour shoveling snow (note that kcalkcal is just a unit of energy). The oxidation of organic nutrients such as glucose during metabolism liberates approximately 3.36kcal of energy per gram of oxygen consumed. If air is 21% oxygen, what volume of air at STP is needed to produce enough energy for the man to clear snow from a 35 minutes of shoveling?arrow_forward19arrow_forward
- What are the major cations found in interstitial fluid?arrow_forwardWhat volume, in milliliters, will 5.3 g of CO2 occupy at STP?arrow_forwardA U-Tube is divided into left and right compartments by a membrane that exclusively allows water to pass through but not any dissolved particles. Initially, the water level is equal on both sides. Then. the following compounds are added to each compartment: Left compartment: 0.50 mol calcium nitrate and 1 mol of glucose(C6H11O6) Right compartment: 0.75 mol maltose (C12H22O11) and 0.75 mol AlPO4 Select the one statement below that is TRUE. a)Water will stop flowing across the membrane b)The right side compartment will have a higher osmotic pressure c)There will be no change in the water level on either side because water always flattens out d)The water level on the right side will be higher e)The water level on the left side will be higherarrow_forward
- Among the following samples of water, which contains the highes K carbonate hardness? O oxygenated water O distilled water O raw wastewater O de-ionized waterarrow_forwardexplain the answerarrow_forwardAt 0 C and 1.00 atm, as much as 0.70 g of O2 can dissolve in 1 L of water. At 0 C and 4.00 atm, how many grams of O2 dissolve in 1 L of water?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax





