
Organic Chemistry - Standalone book
10th Edition
ISBN: 9780073511214
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 25.7, Problem 8P
Locate the isoprene units in each of the monoterpenes, sesquiterpenes, and diterpenes shown in Figure

Diterpenes:
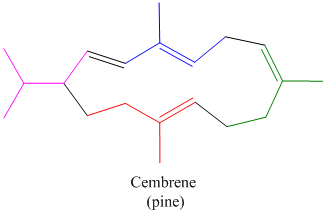
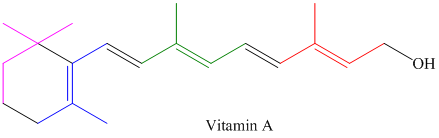
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
is Isoprene ionic or covalent? Why?
What are the differences between terpenes and terpenoids?
Classify the following terpenes, draw the structure, then give the characteristic property and the natural occurrence: gibberellin, limonene, digitonin
Fill in the empty blanks and spaces in the table. The structures of d-galactose and a-D-galactopyranose are in the next picture for reference.
Chapter 25 Solutions
Organic Chemistry - Standalone book
Ch. 25.2 - What fatty acids are produced on hydrolysis of...Ch. 25.3 - Give the structure of the keto acyl-ACP formed...Ch. 25.4 - Prob. 3PCh. 25.4 - Prob. 4PCh. 25.5 - Prob. 5PCh. 25.6 - Write the structural formula and give the IUPAC...Ch. 25.6 - Prob. 7PCh. 25.7 - Locate the isoprene units in each of the...Ch. 25.7 - Prob. 9PCh. 25.9 - Prob. 10P
Ch. 25.9 - Prob. 11PCh. 25.10 - Prob. 12PCh. 25.11 - Prob. 13PCh. 25.11 - Prob. 14PCh. 25.12 - Prob. 15PCh. 25.16 - Prob. 16PCh. 25 - Prob. 17PCh. 25 - Prob. 18PCh. 25 - Prob. 19PCh. 25 - Pyrethrins are a group of naturally occurring...Ch. 25 - Prob. 21PCh. 25 - Prob. 22PCh. 25 - Prob. 23PCh. 25 - Prob. 24PCh. 25 - Prob. 25PCh. 25 - Prob. 26PCh. 25 - Prob. 27PCh. 25 - Prob. 28PCh. 25 - A synthesis of triacylglycerols has been described...Ch. 25 - Prob. 30PCh. 25 - Prob. 31PCh. 25 - Prob. 32PCh. 25 - Prob. 33DSPCh. 25 - Prob. 34DSPCh. 25 - Prob. 35DSPCh. 25 - Prob. 36DSPCh. 25 - Prob. 37DSPCh. 25 - Prob. 38DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Is the attached compound not composed of isoprene units? Locate the isoprene units in each compound that contains them.arrow_forwardWhich isoprene units are connected in a head-to-tail fashion?arrow_forwardGuaiol is a sesquiterpene alcohol found in cannabis and other plants. The structure of guaiol is drawn except for a missing CH3 group. If the isoprene units are joined in a head-to-tail fashion, draw a possible structure for guaiol that contains the additional CH3 group.arrow_forward
- Draw out the structure for lycopene:- Mark each isoprene unit- Where is lycopene found in nature and what health benefits does it provide?arrow_forwardTalk about polystyrene in general (detailed explanation), while writing references cited from itarrow_forwardMark off the isoprene units in lycopene and β-carotene. Can you detect a similarity in the way in which squalene, lycopene, and β-carotene are biosynthesized?arrow_forward
- PVA has an average molar mass of 78,000 g/mol. On average, how many [CH2CHOH] monomer units are in a strand of PVA?arrow_forwardWhat are the possible products of catalytic reforming of normal heptane?arrow_forwardWhich of the following statements is true concerning glucose polymers? A. Starch is a glucose polymer held together by an α-(1,6) linkage. B. Humans do not eat foods that contain cellulose. C. Glucose is not favored in the body in ring form. D. Humans lack the enzyme that can break down the beta linkages in cellulose.arrow_forward
- What is the monomer of PMMA?arrow_forward(a) Locate the isoprene units in lycopene, the red pigment in tomatoes. (b) Which isoprene units are connected in a head-to-tail fashion? (c) Label any other isoprene unit as connected in a head-tohead fashion or a tail-to-tail fashion. (d) Classify lycopene as a monoterpene, sesquiterpene, and so on.arrow_forwardDraw the structure of the terpene limonene and B-Caryophyllene and outline the isoprene units that make up these molecules. .arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY