
ORGANIC CHEM.(LL)-W/OWL V2 >CUSTOM<
9th Edition
ISBN: 9781337034623
Author: McMurry
Publisher: CENGAGE C
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 25.SE, Problem 52AP
Interpretation Introduction
Interpretation:
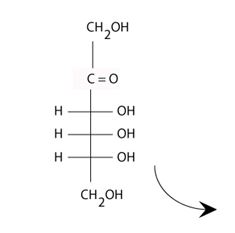
The structure of the D-2-ketohexose psicose is: allitol and altritol.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
A 2-ketohexose is reduced with NaBH4 in CH3OH to form a mixture of D-galactitol and Dtalitol. What is the structure of the 2-ketohexose?
Predict the products obtained when d-galactose reacts with each reagent. (f) excess Ac2O and pyridine (g) excess CH3 I, Ag2O
Identify the sugar in each description. a. An aldopentose that is not d-arabinose forms d-arabinitol when it is reduced with NaBH4. b. A sugar that is not D-altrose forms d-altraric acid when it is oxidized with nitric acid. c. A ketose that, when reduced with NaBH4, forms d-altritol and d-allitol.
Chapter 25 Solutions
ORGANIC CHEM.(LL)-W/OWL V2 >CUSTOM<
Ch. 25.1 - Prob. 1PCh. 25.2 - Prob. 2PCh. 25.2 - Prob. 3PCh. 25.2 - Prob. 4PCh. 25.2 - Prob. 5PCh. 25.3 - Prob. 6PCh. 25.3 - Prob. 7PCh. 25.4 - Prob. 8PCh. 25.4 - Prob. 9PCh. 25.4 - Prob. 10P
Ch. 25.5 - Prob. 11PCh. 25.5 - Prob. 12PCh. 25.5 - Prob. 13PCh. 25.5 - Prob. 14PCh. 25.5 - Prob. 15PCh. 25.6 - Prob. 16PCh. 25.6 - Prob. 17PCh. 25.6 - Prob. 18PCh. 25.6 - Prob. 19PCh. 25.6 - Prob. 20PCh. 25.6 - Prob. 21PCh. 25.6 - Prob. 22PCh. 25.6 - Prob. 23PCh. 25.7 - Prob. 24PCh. 25.8 - Show the product you would obtain from the...Ch. 25.SE - Prob. 26VCCh. 25.SE - Prob. 27VCCh. 25.SE - Prob. 28VCCh. 25.SE - Prob. 29VCCh. 25.SE - Prob. 30MPCh. 25.SE - Prob. 31MPCh. 25.SE - Glucosamine, one of the eight essential...Ch. 25.SE - D-Glicose reacts with acetone in the presence of...Ch. 25.SE - Prob. 34MPCh. 25.SE - Prob. 35MPCh. 25.SE - Prob. 36APCh. 25.SE - Prob. 37APCh. 25.SE - Prob. 38APCh. 25.SE - Prob. 39APCh. 25.SE - Prob. 40APCh. 25.SE - Assign R or S configuration to each chirality...Ch. 25.SE - Prob. 42APCh. 25.SE - Prob. 43APCh. 25.SE - Prob. 44APCh. 25.SE - Prob. 45APCh. 25.SE - Prob. 46APCh. 25.SE - Prob. 47APCh. 25.SE - Prob. 48APCh. 25.SE - Prob. 49APCh. 25.SE - Prob. 50APCh. 25.SE - Prob. 51APCh. 25.SE - Prob. 52APCh. 25.SE - Prob. 53APCh. 25.SE - Prob. 54APCh. 25.SE - Prob. 55APCh. 25.SE - Prob. 56APCh. 25.SE - Prob. 57APCh. 25.SE - Prob. 58APCh. 25.SE - Prob. 59APCh. 25.SE - Prob. 60APCh. 25.SE - Prob. 61APCh. 25.SE - Prob. 62APCh. 25.SE - Prob. 63APCh. 25.SE - D-Mannose reacts with acetone to give a...Ch. 25.SE - Prob. 65APCh. 25.SE - Prob. 66APCh. 25.SE - Prob. 67APCh. 25.SE - Prob. 68APCh. 25.SE - Prob. 69APCh. 25.SE - Prob. 70APCh. 25.SE - Prob. 71AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 1. Carbohydrates classification. 2. Write down the reactions: a) α,D-Glucopyranose + C2H5OH → b) D-Glucose + [Ag(NH3)2]+ → c) D-Glucopyranose + (CH3CO)2 O → d) D-Glucopyranose + CH3I → e) D-Glucose + HNO3 → f) D-Glucose + H2 → g) Lactose formation h) Sucrose hydrolysis 3. Write down the formula of β,D-galactopyranosearrow_forwardA D-aldohexose A is formed from an aldopentose B by the Kiliani–Fischer synthesis. Reduction of A with NaBH4 forms an optically inactive alditol. Oxidation of B forms an optically active aldaric acid. What are the structures of A and B?arrow_forwardWhat products are obtained from the reduction of a. d-idose? b. d-sorbose?arrow_forward
- Predict the products obtained when d-galactose reacts with each reagent. (d) Ag(NH3) 2+ -OH(e) H2, Ni (f) excess Ac2O and pyridinearrow_forwardIdentify the sugar in description. An aldopentose that is not d-arabinose forms d-arabinitol when it is reduced with NaBH4.arrow_forwardWhen D-glucose is reduced with sodium borohydride,optically active glucitol results but when optically D-galactose is reduced ,the product is inactive why?arrow_forward
- Predict the products obtained when d-galactose reacts with each reagent. (h) NaBH4(i) Br2, H2O, then H2O2 and Fe2(SO4)3arrow_forwardPredict the products obtained when d-galactose reacts with each reagent. (c) CH3OH, H + (d) Ag(NH3) 2+ -OH(e) H2, Niarrow_forwardWhat product is formed when D-arabinose is treated with each reagent: (a) H 2 , Pd; (b) Benedict’s reagent?arrow_forward
- Draw the products formed when β-D-galactose is treated with each reagent. a. Ag2O + CH3I b. NaH + C6H5CH2Cl c. The product in (b), then H3O+ d. Ac2O + pyridine e. C6H5COCl + pyridine f. The product in (c), then C6H5COCl + pyridinearrow_forwardIllustrate (hand-drawn) the mechanism of intrahemiacetal or intrahemiketal formation in the following monosaccharides and identify/draw the possible products of these reactions. A. D-talose B. D-fructosearrow_forwardDraw Fischer projections for the product(s) formed by reaction of d-ribose with the following. In addition, state whether each product is optically active or inactive Q. HNO3, warmarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
