
ORGANIC CHEMISTRY SOLUTION MANUAL
16th Edition
ISBN: 9781260036510
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 26, Problem 26.46P
Devise a synthesis of each substituted cyclopropane. Use acetylene
a. 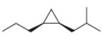 b.
b. 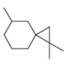
+ enantiomer
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Devise a synthesis of each substituted cyclopropane. Use acetylene (HC=CH) as a starting material in part (a) and cyclohexanone as a starting material in part (b). You may use any other organic compounds and any needed reagents.
Devise a synthesis of each alkene using a Wittig reaction to form the double bond. You may use benzene and organic alcohols having four or fewer carbons as starting materials and any required reagents.
Devise a synthesis of each compound using 1-bromobutane(CH3CH2CH2CH2Br) as the only organic starting material. You may useany other inorganic reagents.
Chapter 26 Solutions
ORGANIC CHEMISTRY SOLUTION MANUAL
Ch. 26 - Prob. 26.1PCh. 26 - Prob. 26.2PCh. 26 - Prob. 26.3PCh. 26 - Prob. 26.4PCh. 26 - Prob. 26.5PCh. 26 - Prob. 26.6PCh. 26 - Prob. 26.7PCh. 26 - Problem 26.8
What starting materials are needed to...Ch. 26 - Prob. 26.9PCh. 26 - Problem 26.10
What reagents are needed to convert...
Ch. 26 - Problem 26.11
What product is formed when each...Ch. 26 - Prob. 26.12PCh. 26 - Problem 26.13
Draw the products formed when each...Ch. 26 - Problem 26.14
What products are formed when ...Ch. 26 - Problem 26.15
Draw the product formed from...Ch. 26 -
What product is formed by ring-closing metathesis...Ch. 26 - Problem 26.17
What starting material is needed to...Ch. 26 - 26.18 In addition to organic halides, alkyl...Ch. 26 - 26.19 What product is formed by ring-closing...Ch. 26 - 26.20 Draw the products formed in each...Ch. 26 - What organic halide is needed to convert lithium...Ch. 26 - 26.22 How can you convert ethynylcyclohexane to...Ch. 26 - 26.23 What compound is needed to convert styrene...Ch. 26 - 26.24 What steps are needed to convert to octane...Ch. 26 - Prob. 26.25PCh. 26 - Prob. 26.26PCh. 26 - 26.27 Draw the products (including stereoisomers)...Ch. 26 - 26.28 Treatment of cyclohexene with and forms...Ch. 26 - Prob. 26.29PCh. 26 - 26.30 What starting material is needed to prepare...Ch. 26 - Prob. 26.31PCh. 26 - Prob. 26.32PCh. 26 - When certain cycloalkenes are used in metathesis...Ch. 26 - 26.34 Draw the products formed in each reaction.
...Ch. 26 - Prob. 26.35PCh. 26 - Draw a stepwise mechanism for the following...Ch. 26 - Sulfur ylides, like the phosphorus ylides of...Ch. 26 - Although diazomethane is often not a useful...Ch. 26 - Prob. 26.39PCh. 26 - Prob. 26.40PCh. 26 - Prob. 26.41PCh. 26 - Prob. 26.42PCh. 26 - 26.43 Devise a synthesis of each compound using a...Ch. 26 - 26.44 Devise a synthesis of each compound from...Ch. 26 - 26.45 Devise a synthesis of each compound from...Ch. 26 - 26.46 Devise a synthesis of each substituted...Ch. 26 - Biaryls, compounds containing two aromatic rings...Ch. 26 - Prob. 26.48PCh. 26 - 26.49 Draw the product formed from the...Ch. 26 - Prob. 26.50PCh. 26 - 26.51 Devise a synthesis of each of the following...Ch. 26 - Prob. 26.52PCh. 26 - 26.53 The following conversion, carried out in the...Ch. 26 - Prob. 26.54PCh. 26 - 26.55 Dimethyl cyclopropanes can be prepared by...Ch. 26 - Prob. 26.56P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Devise a synthesis of attached alkene using a Wittig reaction to form the double bond. You may use benzene and organic alcohols having four or fewer carbons as starting materials and any required reagents.arrow_forwarda. Draw the two chair forms for compound A, being careful to show the stereoisomergiven above. You may use shorthand R in your drawings. Indicate the MAJOR and MINORconformations b. Using curved-arrow convention and the correct chair form of Compound A, show themechanism for the formation of alkene C from Compound A. You may use B: to indicatethe base.arrow_forwardA key step in the synthesis of β-vetivone, a major constituent of vetiver, a perennial grass found in tropical and subtropical regions of the world, involved the reaction of compound A and dihalide B with two equivalents of LDA to form C. Draw a stepwise mechanism for this reaction. β-Vetivone contains a spiro ring system—that is, two rings that share a single carbon atom.arrow_forward
- Uridine monophosphate (UMP) is one of the four nucleotides that compose RNA, the nucleic acid that translates the genetic information of DNA into proteins needed by cells for proper function and development. A key step in the synthesis of UMP is the SN1 reaction of A with B to form C, which is then converted to UMP in one step. Draw a stepwise mechanism for this SN1 reaction.arrow_forwardIn addition to using CHX3 and base to synthesize dihalocarbenes (Section 26.4), dichlorocarbene (:CCl2) can be prepared by heating sodium trichloroacetate. Draw a stepwise mechanism for this reaction.arrow_forwardDraw a stepwise mechanism for the following substitution. Explain why 2-chloropyridine reacts faster than chlorobenzene in this type of reaction.arrow_forward
- Answer the following questions about curcumin, a yellow pigment isolated from turmeric, a tropical perennial in the ginger family and a principal ingredient in curry powder.a.In Chapter 11, we learned that most enols, compounds that contain a hydroxy group bonded to a C=C, are unstable and tautomerize to carbonyl groups. Draw the keto form of the enol of curcumin, and explain why the enol is more stable than many other enols. b.Explain why the enol O—H proton is more acidic than an alcohol O—H proton. c. Why is curcumin colored? d.Explain why curcumin is an antioxidant.arrow_forwardConsider the tetracyclic compound with rings labeled A–D. (a) Which ring is the most reactive in electrophilic aromatic substitution? (b) Which ring is the least reactive in electrophilic aromatic substitution?arrow_forwardDraw the major organic product or give the reagents needed to complete each.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY