
Concept explainers
Answer the following questions for this
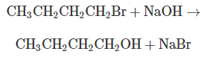
a. What is the rate expression for the reaction?
b. Draw the reaction profile for the reaction Label all parts Assume that the products are lower in energy than the reactants
c. What is the effect on the
d. What is the effect on the rate of the reaction of halving the concentration of sodium hydroxide?
Want to see the full answer?
Check out a sample textbook solution
Chapter 27 Solutions
Selected Solutions Manual For General Chemistry: Principles And Modern Applications
Additional Science Textbook Solutions
CHEMISTRY-TEXT
Chemistry (7th Edition)
Chemistry
Principles of Chemistry: A Molecular Approach (3rd Edition)
Principles of General, Organic, Biological Chemistry
Chemistry: Structure and Properties (2nd Edition)
- 3-Bromo-1-butene and 1-bromo-2-butene undergo SN1 reaction at nearly the same rate, even though one is a secondary halide and the other is primary. Explain.arrow_forward7. Choose the best reagent from the list below for carrying out each transformation. Place the letter of the reagent in the blank to the left of the reaction. (see attached screenshot). a. 1. O3 2. Zn, H3O+ b. 1. BH3, THF 2. H2O2, NaOH, H2O c. H2O, H2SO4, heat d. 1. OsO4 2. NaHSO3, H2O e. KMnO4, acid f. 1. Hg(OAc)2, H2O 2. NaBH4arrow_forward1. For the following reaction, what is the rate law ? (image) 2. is the following nucleophile strong or weak HO^- a) strong b) weak c) not a nucleophile 3) what set of reaction conditions should favor an SN2 reaction on 2-bromo-3-methylbutane a) weak nucleophile in a protic solvent b) weal nucleophile in aprotic solvent c) strong nucleophile in a protic solvent d) strong nucleophile in a aprotic solventarrow_forward
- 1. Which of the following is the strongest nucleophile? Refer to the diagram below with an MeOH, MeSH, etc. 2. Which of the following is true about the stereochemistry of SN1 reaction?a.retention of configuration at the electrophilic centerb.50:50 mixture of retention and inversion of configuration at the electrophilic centerc.inversion of configuration at the electrophilic centerd. slightly more inversion than retention at the electrophilic centerarrow_forwardConsider the reaction of (R)-2-chloro-3-methylbutane with sodium iodide to form aproduct.1(a) Draw the reaction scheme with the correct stereochemistry (reactant + NaI → product+ NaCl). Circle the nucleophile and draw a rectangle around the electrophile. 1(b) What is the symbol used for mechanism shown in 1(a)?1(c) If the sodium iodide was replaced with sodium hydroxide, the product is anALKENE. Draw a reaction MECHANISM to show how this happens.1(d) Draw the reaction energy diagram for the reaction in 1(c) and label the activationenergy.1(e) Using any alcohol with five carbons, and any carboxylic acid with six carbons, draw areaction to show how we would make an ester. 1(f) Describe the practical on esters.arrow_forwardWhat kind of reagent is KmnO4? Draw the possible reactions of this reagent with the specific compounds tested in the following reaction?arrow_forward
- For which reaction mechanisms—SN1, SN2, E1, or E2—are each of the following statements true? A statement may be true for one or more mechanisms. a.The mechanism involves carbocation intermediates. b.The mechanism has two steps. c.The reaction rate increases with better leaving groups. d.The reaction rate increases when the solvent is changed from CH3OH to (CH3)2SO. e.The reaction rate depends on the concentration of only the alkyl halide. f.The mechanism is concerted. g.The reaction of CH3CH2Br with NaOH occurs by this mechanism. h.Racemization at a stereogenic center occurs. i.Tertiary (3°) alkyl halides react faster than 2° or 1° alkyl halides. j.The reaction follows a second-order rate equation.arrow_forwardFor which reaction mechanisms—SN1, SN2, E1, or E2—are each of the following statements true? A statement may be true for one or more mechanisms.a. The mechanism involves carbocation intermediates.b. The mechanism has two steps.c. The reaction rate increases with better leaving groups.d. The reaction rate increases when the solvent is changed from CH3OH to (CH3)2SO.e. The reaction rate depends on the concentration of the alkyl halide only.f. The mechanism is concerted.g. The reaction of CH3CH2Br with NaOH occurs by this mechanism.h. Racemization at a stereogenic center occurs.i. Tertiary (3°) alkyl halides react faster than 2° or 1° alkyl halides.j. The reaction follows a second-order rate equation.arrow_forwardDraw the products of the three step reaction sequence shown below. Ignore inorganic byproducts. If the reaction results in a mixture of ortho and para isomers, draw only the para-product. Select to Draw NO₂ 1. LiAlH4 2. H₂O* Cl₂ AICI 3 Select to Draw CH3C(O)CI Select to Drawarrow_forward
- How does changing the alkyl halide from CH3CH2Br to (CH3)2CHBr affect the rate of an E2 reaction?arrow_forwardHow does changing the halide from (CH3)3CBr to CH3CH2CH2Br affect the rate of an E1 reaction?arrow_forwardChoose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) Br2, FeBr3 Na/NH3, -33 degrees C NBS, light KMnO4, H3O+ Mg metal, ether KOH, EtOH, heatarrow_forward

