
Interpretation:
For the
Concept introduction:
Polymer may contain one or more than one functional group in its structure.
Answer to Problem 15P
Solution: Acetylene.
Explanation of Solution
The repeating units of a polymer are known as monomers. The process by which respective monomers combine to form polymers is known as polymerization. The polymer formed by the simple addition reactions of monomers without the generation of by-products are known as
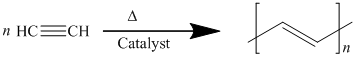
This is a repeating unit of polyacetylene whose IUPAC name is polyethylene. The monomer of this polymer is acetylene with molecular formula
The monomer is acetylene.
Want to see more full solutions like this?
Chapter 28 Solutions
Organic Chemistry (Binghampton University)
- The following polymer may be used as polymer binders for coatings; please write down the structure of the monomer(s) that should be used to synthesize the polymerarrow_forwardDraw a structure of the polymer formed in the following reactionarrow_forwardDraw the structure of the monomer or monomers used to synthesize the following polymers, and indicate whether each is a chain-growth polymer or a step-growth polymer.arrow_forward
- Commercial polyisobutylene (PIB or butyl rubber) is synthesized via cationic polymerization. Explain why it cannot be made through free radical or anionic polymerizationarrow_forwardShow the structure of the polymer that results from heating the following diepoxide and diamine:arrow_forwardRadical polymerization of styrene gives a linear polymer. Radical polymerization of a mixture of styrene and 1,4-divinylbenzene gives a cross-linked network polymer of the type shown in Figure 29.1. Show by drawing structural formulas how incorporation of a few percent of 1,4-divinylbenzene in the polymerization mixture gives a cross-linked polymer.arrow_forward
- A sample of polystyrene is composed of a series of fractions of different sized molecules. a)Calculate the number average and weight average molecular weights of this sample as well as the PDI. Comment on the PDI – is it polydispersed? b)How would adding styrene oligomer change the average molecular weights? Added amount is 5 wt. % of polymer mass and M = 1,000 g/mol. c)What would be the potential polymerisation pathways you may follow to polymerise this polymer d)What would happen if you fractionate the polymer to remove all fractions above 20,000 Da and 50% of any fraction above 15,000 Da? Recalculate the Mn, Mw, and PDI for this new system. e)What type of polymeric materials does this distribution of Mw for the new system corresponds to? Would it be likely solid or liquid at room temperature and atmospheric pressure?arrow_forwardPoly(butylene terephthalate) is a hydrophobic plastic material widely used in automotive ignition systems. Suggest what monomers might be used to synthesize this polymer and how the polymerization might beaccomplished.arrow_forwardPoly(ethylene terephthalate) is a linear polyester that can be synthesized from the condensation polymerization between terephthalic acid and ethylene glycol, as shown previously in Equation 26-27. What alcohol would you react with terephthalic acid to produce a branched polyester? Draw a portion of the resulting polymer that shows a main chain and one branching point.arrow_forward
- QUESTION1: (this is the full question however can you answer part c,e and f) Methyl methacrylate is polymerized by free radical polymerization at 70 oC to form long polymer chains.(a) Draw the chemical structure of methyl methacrylate, define its molecular formula and molecular weight (in g/mol). (b) If benzoyl peroxide is used as the initiator for the polymerization of methyl methacrylate please write the chemical structures of the compounds formed during the initiation step ofthe polymerization. What would happen if the reaction is not heated at 70 oC? (c) Write the chemical structures of the polymers formed during the propagation and termination steps of the polymerization. (d) Write the trade name of the polymer formed by the polymerization of methyl methacrylate and two of its common applications. (e) If a polymer with degree of polymerization 100 is synthesized during the reaction, determine its molecular weight (in g/mol). (f) If from the polymerization of methyl methacrylate a…arrow_forwardWhat is the role of benzoyl peroxide in addition polymerisation of alkenes? Explain its mode of action with the help of an example.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co


