
ORGANIC CHEMISTRY(LL)-W/ACCESS >CUSTOM<
10th Edition
ISBN: 9781259917196
Author: Carey
Publisher: MCG CUSTOM
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 28, Problem 23P
(a)
(b) 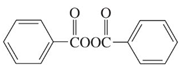
(c)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
7cd (JUST SIMPLY ANSWER THE QUESTION BELOW)
1. Why are olefins (alkanes) good monomers for polymerization reaction?
2. "Co-polymers" consist of two different monomers ("A" and "B") joined in an alternating fashion (ABABAB...). Block co-polymers also consist of two different monomers, but in this case, blocks of polymer containing only A units are joined to blocks of polymer containing only B units (AAAAABBBBB...). How block co-polymers are synthesized?
Macromolecules can also be formed from cyclic monomers such as cyclic hydrocarbons, ethers, esters, amides, siloxanes, and sulfur (eight-membered ring). Thus, ring-opening polymerization is of particular interest, since macromolecules of almost any chemical structure can be prepared. Choose two of the above type polymers to explain the mechanism and chemical reaction of polymerization process.
Olefin polymerization is responsible for producing over 100 million tons per year of plastic material. (a) Briefly describe the type of catalysts used in the transition metal catalysed homogeneous polymerization reactions. (b) How are the catalysts activated? (c) Draw the active catalyst, and indicate how the polymer chain is propagated. (d) Suggest a possible chain termination step. (e) Certain single-site catalysts are capable of selectively forming different types of polypropylene. Describe these different types of polymer, and the characteristics of the catalysts that form them.
Chapter 28 Solutions
ORGANIC CHEMISTRY(LL)-W/ACCESS >CUSTOM<
Ch. 28.2 - Prob. 1PCh. 28.3 - Prob. 2PCh. 28.3 - Prob. 3PCh. 28.4 - Prob. 4PCh. 28.7 - Problem 28.5 (a) Write a chemical equation for the...Ch. 28.7 - Prob. 6PCh. 28.8 - Prob. 7PCh. 28.9 - Problem 28.8 How will the average chain length of...Ch. 28.9 - Prob. 9PCh. 28.11 - Prob. 10P
Ch. 28.11 - Prob. 11PCh. 28.12 - Prob. 12PCh. 28.13 - Prob. 13PCh. 28.14 - Prob. 14PCh. 28 - Prob. 15PCh. 28 - Prob. 16PCh. 28 - Prob. 17PCh. 28 - Prob. 18PCh. 28 - Prob. 19PCh. 28 - Prob. 20PCh. 28 - Of the following monomers, which one would undergo...Ch. 28 - Prob. 22PCh. 28 - Polymerization of styrene can occur by a...Ch. 28 - Styrene undergoes anionic polymerization at a...Ch. 28 - Prob. 25PCh. 28 - Prob. 26PCh. 28 - Prob. 27PCh. 28 - Prob. 28PCh. 28 - Prob. 29PCh. 28 - Prob. 30PCh. 28 - Prob. 31PCh. 28 - Prob. 32DSPCh. 28 - Prob. 33DSPCh. 28 - Prob. 34DSPCh. 28 - Prob. 35DSPCh. 28 - Prob. 36DSPCh. 28 - Prob. 37DSPCh. 28 - Prob. 38DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In a non-acid catalyzed step-growth polymerization between A-A and B-B monomers, the A groups are titratable by aqueous base (1 N) and [A-A] [B-B) = 6 mol.kg-1. a) What is the DP if it takes 100 mL of base to neutralize the reaction mixture at 700 minutes of reaction time (assuming the initial total weight 500 g)?...arrow_forwardPolyethylene is produced by addition polymerization process called Free radical polymerization, which of the following steps is not part of this process: a) initiation b) termination c) propagationarrow_forwardWhat is the first step in the cationic polymerization of alkenes?arrow_forward
- What is the main reaction and mechanism for the polymerization of styrene? What are the side reactions?arrow_forwardWhich of the following monomers is suitable for condensation polymerization? A. Diacid and Diamine B. Vinyl chloride C. Ethane D. Propenearrow_forwardShow the differences between the chain-growth and step-growth mechanisms of polymerization.arrow_forward
- Which of these is a requirement of monomers in order to undergo addition polymerization? a. The presence of an oxygen atom. b. The presence of a carbon to carbon double bond. c. The presence of a carbon to carbon triple bond. d. The presence of a lone pair.arrow_forward"Co-polymers" consist of two different monomers ("A" and "B") joined in analternating fashion (ABABAB...). Block co-polymers also consist of twodifferent monomers, but in this case, blocks of polymer containing only Aunits are joined to blocks of polymer containing only B units(AAAAABBBBB...). How block co-polymers are synthesized?arrow_forwardOne common type of cation exchange resin is prepared by polymerization of a mixture containing styrene and 1,4-divinylbenzene . The polymer is then treated with concentrated sulfuric acid to sulfonate a majority of the aromatic rings in the polymer. Q.) Explain how this sulfonated polymer can act as a cation exchange resinarrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you

 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning


General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning
CBSE Class 12 Chemistry || Polymers || Full Chapter || By Shiksha House; Author: Best for NEET;https://www.youtube.com/watch?v=OxdJlS0xZ0Y;License: Standard YouTube License, CC-BY