
Concept explainers
(a)
Interpretation: The relationship between ![]() and
and ![]() is to be stated.
is to be stated.
Concept introduction: The anomers are cyclic monosaccharides, which differ in configuration at one stereogenic centre. These carbon atoms are called anomeric centre.
Answer to Problem 28.42P
The compounds ![]() and
and ![]() are epimers.
are epimers.
Explanation of Solution
The structures of compounds ![]() and
and ![]() are,
are,
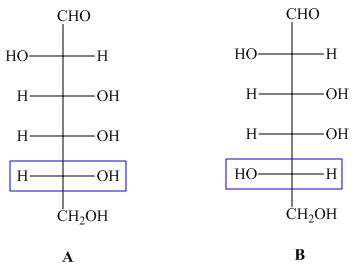
Figure 1
In the given structures, the configuration at ![]() is different. Hence, the compounds
is different. Hence, the compounds ![]() and
and ![]() are epimers.
are epimers.
The compounds ![]() and
and ![]() are epimers.
are epimers.
(b)
Interpretation: The relationship between ![]() and
and ![]() is to be stated.
is to be stated.
Concept introduction: The anomers are cyclic monosaccharides, which differ in configuration at one stereogenic centre. These carbon atoms are called anomeric centre. The compounds which are neither mirror images nor supperimposable on each other are known as diastereomer.
Answer to Problem 28.42P
The compound ![]() and
and ![]() are diastreomers but not epimers.
are diastreomers but not epimers.
Explanation of Solution
The structures of given compound ![]() and
and ![]() are,
are,
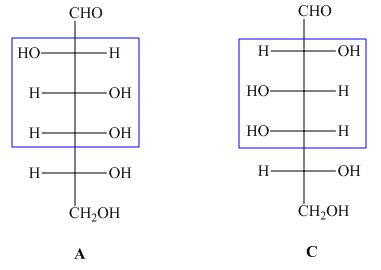
Figure 2
In the given structures, the compound ![]() is not mirror image of compound
is not mirror image of compound ![]() and the configuration of more than is different. Hence, the compound
and the configuration of more than is different. Hence, the compound ![]() and
and ![]() are diastreomers but not epimers.
are diastreomers but not epimers.
The compound ![]() and
and ![]() are diastreomers but not epimers.
are diastreomers but not epimers.
(c)
Interpretation: The relationship between ![]() and
and ![]() is to be stated.
is to be stated.
Concept introduction: The compounds which are mirror images as well as non-supperimposable on each other. These compounds are known as enantiomers.
Answer to Problem 28.42P
The compound ![]() and
and ![]() are enantiomers.
are enantiomers.
Explanation of Solution
The structures of given compound ![]() and
and ![]() are,
are,
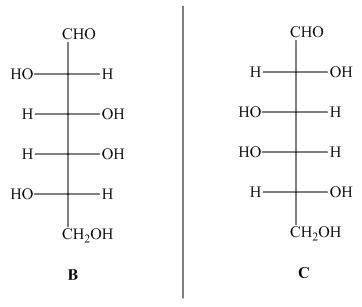
Figure 3
In the given structures, the compound ![]() is mirror image of compound
is mirror image of compound ![]() . Hence, the compound
. Hence, the compound ![]() and
and ![]() are enantiomers.
are enantiomers.
The compound ![]() and
and ![]() are enantiomers.
are enantiomers.
(d)
Interpretation: The relationship between ![]() and
and ![]() is to be stated.
is to be stated.
Concept introduction: The compounds which have same molecular formula but differ in connectivity of the substituents. These compounds are known as constitutional isomers.
Answer to Problem 28.42P
The compound ![]() and
and ![]() are constitutional isomers.
are constitutional isomers.
Explanation of Solution
The structures of given compound ![]() and
and ![]() are,
are,
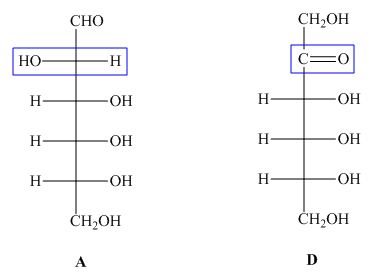
Figure 4
In the given structures, the molecular formula of ![]() is same to the molecular formula of
is same to the molecular formula of ![]() but the connectivity of substituents at
but the connectivity of substituents at ![]() is different. Hence, the compound
is different. Hence, the compound ![]() and
and ![]() are constitutional isomers.
are constitutional isomers.
The compound ![]() and
and ![]() are constitutional isomers.
are constitutional isomers.
(e)
Interpretation: The relationship between ![]() and
and ![]() is to be stated.
is to be stated.
Concept introduction: The anomers are cyclic monosaccharides, which differ in configuration at one stereogenic centre. These carbon atoms are called anomeric centre. The compounds which are neither mirror images nor supperimposable on each other are known as diastereomer.
Answer to Problem 28.42P
The compounds ![]() and
and ![]() are diastreomers but not epimers.
are diastreomers but not epimers.
Explanation of Solution
The substituents on a carbon are present above the ring in Haworth projection indicates that these bonds are above the plane (i.e. either equatorial or axial position) in chair form. The structures of given compound ![]() and
and ![]() are,
are,
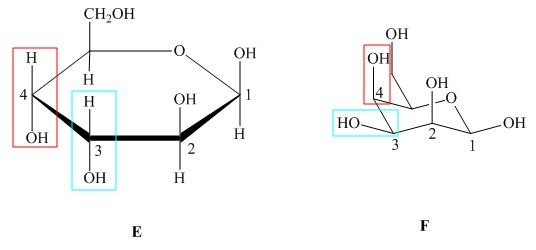
Figure 5
In the given structures, the configuration at ![]() is different which shows that the compound
is different which shows that the compound ![]() is not mirror image of compound
is not mirror image of compound ![]() and. Hence, the compound
and. Hence, the compound ![]() and
and ![]() are diastreomers but not epimers.
are diastreomers but not epimers.
The compounds ![]() and
and ![]() are diastreomers but not epimers.
are diastreomers but not epimers.
Want to see more full solutions like this?
Chapter 28 Solutions
ORGANIC CHEMISTRY
- Indicate the relationship between each pair. Choose from: configurational stereoisomers,conformers, constitutional isomers, or different formulas (Each term is used at least twice.)arrow_forwardHow are the compounds in each pair related to each other? Are they identical, enantiomers, diastereomers, constitutional isomers, or not isomers of each other? a. b. H НО H 'OH H and and H H H HO OHarrow_forwardHow are the compounds in each pair related to each other? Are they identical, enantiomers, diastereomers, constitutional isomers, or not isomers of each other? CH3 a. b. C. d. e. f. CH3 HC. HO I CI CH3 CH3 Br H C and and CHO H OH and and and and H CI OHC HC HO CI CBr CH3 OH g. h. j. Br I. HO • k. H and •pt-pt. OH BrCH₂ H H CH3 H HỌ CH3 "Br H and CCH₂OH CH3 and and CH₂Br and CH3 H Br HC Br H. and HO CH3- CH3 CH3 H HOCH₂ H BrCH₂ Br H CH3 CH3 _CH_OH H OH Harrow_forward
- Drawn are four isomeric dimethylcyclopropane. a. How are the compounds in each pair related (enantiomers, diastereomers,constitutional isomers): A and B; A and C; B and C; C and D?b. Label each compound as chiral or achiral.c. Which compounds, alone, would be optically active?d. Which compounds have a plane of symmetry?e. Which of the compounds are meso compounds?f. Would an equal mixture of compounds C and D be optically active? Whatabout an equal mixture of B and C?g. How many stereogenic centers are there for each compound?arrow_forwardSaquinavir (trade name Invirase) belongs to a class of drugs called protease inhibitors, which are used to treat HIV (human immunodeficiency virus). OH CONH2 O saquinavir Trade name: Invirase NH a. Locate all stereogenic centers in saquinavir, and label each stereogenic center as R or S. b. Draw the enantiomer of saquinavir. c. Draw a diastereomer of saquinavir. d. Draw a constitutional isomer that contains at least one different functional group.arrow_forwardHow is each compound (B–D) related to A? Choose from enantiomers, diastereomers, identical molecules, constitutional isomers, or not isomers of each other.arrow_forward
- Which of the following pairs of structures represent a pair of enantiomers? a. b. C. d. O a O b Oc C Od H3C... H Br C HC NC H3CH₂C COOH H... C CN CH3 Br HC H₂N CH3 OH COOH CN HC-CH3 Br Br H.C. HOOC OH H... C H₂C CN H3C C H₂N CH₂CH3 COOH Harrow_forwardClassify each pair of compounds as constitutional isomers or stereoisomers. a. and b. and OH с. and d. andarrow_forwardLocate the stereogenic centers in each compound and draw the enantiomer. Exemestane (trade name Aromasin) is used to treat breast cancer, and zanamivir is used to treat and prevent influenza. a. I H Ill exemestane Oarrow_forward
- 8) Identify enantiomers. C E A CI H ICL F CI H Cle H. CI H. CI H CI CI H. F H. 'F 'F H. F H H. F H H H. A) B and D, A and C B) B and D C) A and B, A and D, B and C, C and D D) B and D, A and B, A and D, B and C, C and D E) A and Carrow_forwardConsider the ball-and-stick models A–D. How is each pair of compounds related: (a) A and B; (b) A and C; (c) A and D; (d) C and D? Choose from identical molecules, enantiomers, or diastereomers.arrow_forwardExplain each statement by referring to compounds A-E. он он HO .OH CI OH A в E a. A has a mirror image but no enantiomer. b. B has an enantiomer and no diastereomer. c. C has both an enantiomer and a diastereomer. d. D has a diastereomer but no enatiomer. e. E has a diastereomer but no enantiomer. E..arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


