
(a)
Interpretation: The product of given reaction has to be drawn.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or brocken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
There are mainly three types of pericyclic reactions,
- 1) Electrocyclic reactions
- 2) Cycloaddition reactions
- 3) Sigmatropic reactions
In an electrocyclic reaction “one new sigma- bond is formed or brocken.”
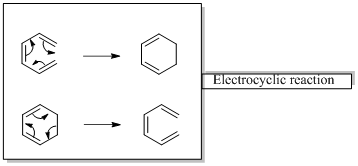
Woodward –Hoffmann rules are the set of rules used to vindicate or predict certain aspects of the stereo chemical outcome and activation energy of pericyclic reactions.
Woodward – Hoffmann rules for Electrocyclic reactions are listed below
A photochemical reaction takes place when a reactant absorbs light and a thermal reaction takes place without the absorption of light.
Woodward – Hoffmann rules for the configuration of electrocyclic reactions are,
(b)
Interpretation: The product of given reaction has to be drawn.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or brocken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
There are mainly three types of pericyclic reactions,
- 1) Electrocyclic reactions
- 2) Cycloaddition reactions
- 3) Sigmatropic reactions
In an electrocyclic reaction “one new sigma- bond is formed or brocken.”

Woodward –Hoffmann rules are the set of rules used to vindicate or predict certain aspects of the stereo chemical outcome and activation energy of pericyclic reactions.
Woodward – Hoffmann rules for Electrocyclic reactions are listed below
A photochemical reaction takes place when a reactant absorbs light and a thermal reaction takes place without the absorption of light.
Woodward – Hoffmann rules for the configuration of electrocyclic reactions are,
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
CHEM 262 ORG CHEM EBOOK DIGITAL DELIVERY
- Which of the synthetic procedures shown in Image 31 would carry out the following transformation? A. b B. c C. a D. darrow_forwardMarkovnikov's rule can best be explained by considering; A. the number of hydrogen atoms attached to each of the double bond carbons atoms. B. the number of alkyl groups attached to each of the double bond carbons atoms. C. the spatial arrangement of the groups around the double bond. D. the relative stabilities of the carbocations that can be formed from each of the double bond carbon atoms.arrow_forwardProvide the missing starting material, reactant or product. Show appropriate stereochemistry. a) b)arrow_forward
- Draw the major organic product for the reaction shown. N. H. HCI, cat.arrow_forwardWhich alkene has (E) configuration? a. b. C. d.arrow_forwardOrganometallic reagents are characterized by a.) polar bonds b.) carbon bonded to an electropositive element c.) carbon behaving as a free carbanion d.) none of thesearrow_forward
- What other alkene is also formed along with Y in Sample Problem 9.3? What alkenes would form from X if no carbocation rearrangement occurred?arrow_forwardWhat is the major organic product obtained from the following reaction? A, B, C, or D?arrow_forwardWhat is the major organic product obtained from the following reaction under low temperature conditions? A, B, C, or D?arrow_forward
- What specific rearrangement occurred in this reactionarrow_forwardWhat name reaction would result to the formation of the cyclohexene structure below? R .CO̟Et A. Diels-Alder reaction B. Wittig Reaction C. Friedel-Craft reaction D. Claisen Condensationarrow_forwardWhat are the major organic products from each reaction??arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
