
CHEM 262 ORG CHEM EBOOK DIGITAL DELIVERY
8th Edition
ISBN: 2818440043505
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 28.5, Problem 13P
- a. Draw the product of the following reaction:
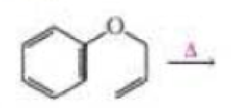
- b. If the terminal sp2 carbon of the substituent attached to the benzene ring is labeled with 14C, where will the label be in the product?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
6. Complete the following reduction reactions, Draw the structure of the product. Name tne
reactant and product.
a)
Cty-CH-C=0
Ni
+ Hs
It
CH3
b) o-と-CH--cng
Ni
Ni
c)
CHy-CH-CHy
+ Hz
CHz
d)
CH
+ Hz
7. Complete the following reactions for the formation of hemiacetals. Draw the structure of the
product. Name the reactants.
a)
acid
Cら-と-A
+ Ho-CHCH
Sつっつ (。
+ Ho-CH3
acid
Ho-CH-CH, + C-CH-C-C-CH d
CH3
c)
Draw the product of the following reaction. If the terminal sp2 carbon of the substituent attached to the benzene ring is labeled with 14C, where will the label be in the product?
a. What five-carbon alkene forms the same product whether it reacts with HBr in the presence of a peroxide or with HBr in the absence of a peroxide?b. Draw the structures of four six-carbon alkenes that form the same product, whether they react with HBr in the presence of a peroxide or with HBr inthe absence of a peroxide.
Chapter 28 Solutions
CHEM 262 ORG CHEM EBOOK DIGITAL DELIVERY
Ch. 28.1 - Prob. 1PCh. 28.2 - Prob. 2PCh. 28.2 - Prob. 3PCh. 28.2 - Give a molecular orbital description for each of...Ch. 28.3 - Prob. 5PCh. 28.3 - Prob. 6PCh. 28.3 - Prob. 7PCh. 28.3 - Prob. 8PCh. 28.4 - Prob. 10PCh. 28.4 - Prob. 11P
Ch. 28.5 - Prob. 12PCh. 28.5 - a. Draw the product of the following reaction: b....Ch. 28.5 - Prob. 14PCh. 28.5 - Prob. 15PCh. 28.5 - Prob. 17PCh. 28.5 - Prob. 18PCh. 28.6 - Prob. 19PCh. 28.6 - Explain why the hydrogen and the methyl...Ch. 28.6 - Chorismate mutase is an enzyme that promotes a...Ch. 28.7 - Convince yourself that the TE-AC method for...Ch. 28 - Draw the product of each of the following...Ch. 28 - Draw the product of each of the following...Ch. 28 - Prob. 25PCh. 28 - Show how norbornance can be prepared from...Ch. 28 - Prob. 27PCh. 28 - Prob. 28PCh. 28 - Draw the product of each of the following...Ch. 28 - Prob. 30PCh. 28 - Prob. 31PCh. 28 - Prob. 32PCh. 28 - Prob. 33PCh. 28 - When the following compound is heated, a product...Ch. 28 - Prob. 35PCh. 28 - Propose a mechanism for the following reaction:Ch. 28 - Prob. 37PCh. 28 - Prob. 38PCh. 28 - Prob. 39PCh. 28 - Prob. 40PCh. 28 - If isomer A is heated to about 100 C, a mixture of...Ch. 28 - Propose a mechanism for the following reaction:Ch. 28 - Prob. 43PCh. 28 - A student found that heating any one of the...Ch. 28 - Prob. 45PCh. 28 - Prob. 46PCh. 28 - Prob. 47P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 35) Draw the major product of the following reaction: 1. R₂BH 2. H₂O₂, OH -C=C-Harrow_forward1. Draw the possible products from the following reaction. io dioxolane + KOH ?arrow_forwardDraw the expected substitution products for the reaction of (R)-2-bromopentane with the following reagents: OH ? Br (strong nucleophile) H₂O ? (weak nucleophile) BI...arrow_forward
- M8. draw the products formed in the following transformations A-Carrow_forward5. What are the products formed when each alkene below is treated first with KMNO4 and KOH in cold water? Are the first products meso or formed as a racemic mixture? What happens when the products of the first step are treated with H2SO4?arrow_forward4. What are the products obtained from the following elimination reaction? Indicate the major product. CH;CH2CCH3 H2O CI 5. a) Determine the major product that is formed when the alkyl halide reacts with a hydroxide ion in an elimination reaction. CH;CH,CH,CH,CCH3 Br b) For the major elimination product obtained in 5a), which stereoisomer (cis or trans) is obtained in greater yield? Draw the two isomers and provide the names of the compounds.arrow_forward
- 6. Draw the reagent that converts the alkene below to the epoxide (oxirane) c=CH2 CH2 H,CH,C° H3CH,C'arrow_forwardWhich is the product of the following reaction? о) C) + CHснинг инск сн он NCH CH. b) ? NH₂ CH CH. нсниarrow_forward4. Draw the products for the following reaction. Answer the questions regarding the reaction. H₂O₂ NaOH A. Which product do you think will be the favored product? Draw it here. B. What do you think will happen if I change the tert-butyl group to a methyl group? a. The ratio of stereoisomers will not change c. The ratio of stereoisomers will go down b. The ratio of stereoisomers will go up d. The ratio of stereoisomers will be equal C. What do you think will happen if move the tert-butyl down to carbon 6? a. The ratio of stereoisomers will not change c. The ratio of stereoisomers will go down b. The ratio of stereoisomers will go up d. The ratio of stereoisomers will be equalarrow_forward
- 8. Draw the major reaction products for the following: a) CH=CH2 Br2 ?arrow_forwardDraw the products of each reaction and indicate the stereochemistry at any stereogenic centers. CH,CH,OH CH;COCI pyridine a. OH C. CH5 COOH H* CH3 CH3 Br NaCN d. CH. HD "NH2 (2 equiv)arrow_forwardDraw the products of the following reaction, then upload all drawings to the question at the end. НО CH₂ CH, ОНarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License