
Concept explainers
(a)
Energy level diagram for one-dimensional box.
(a)
Answer to Problem 43P
The energy level diagram is given below:
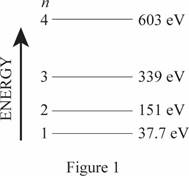
Explanation of Solution
The length of the box is
Write the expression for energy of one dimensional box
Here,
Conclusion
Substitute
Substitute
Substitute
Substitute
The energy level diagram is given below:
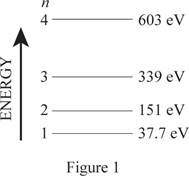
(b)
The wavelength of emitted photons during transitions in these four levels.
(b)
Answer to Problem 43P
The frequencies of the emitted photon are
Explanation of Solution
The minimum frequency for photoemission corresponds to the cutoff wavelength.
Write the expression for wavelength
Here,
Conclusion
For transition,
Substitute
For transition,
Substitute
For transition,
Substitute
For transition,
Substitute
For transition,
Substitute
For transition,
Substitute
Thus, the frequencies of the emitted photon are
Want to see more full solutions like this?
Chapter 28 Solutions
Principles of Physics
- When a hydrogen atom is in its ground state, what are the shortest and longest wavelengths of the photons it can absorb without being ionized?arrow_forwardWhat is the de Brogue wavelength of an electron that is accelerated from rest through a potential difference of 20 keV?arrow_forwardWhy are X-rays emitted only for electron transitions to inner shells? What type of photon is emitted for transitions between outer shells?arrow_forward
- What is the de Brogue wavelength of a proton whose kinetic energy is 2.0 MeV? 10.0 MeV?arrow_forwardCan the de Brogue wavelength of a particle be known exactly?arrow_forwardIf the work function of a metal is 3.2 eV, what is the maximum wavelength that a photon can have to eject a photoelectron from this metal surface?arrow_forward
- What is the de Brogue wavelength of an electron travelling at a speed of 5.0106 m/s ?arrow_forwardA quantum mechanical oscillator vibrates at a frequency of 250.0 THz. What is the minimum energy of radiation it can emit?arrow_forwardWhat is the maximum kinetic energy of photoelectrons ejected from sodium by the incident radiation of wavelength 450 nm?arrow_forward
 Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax
University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill
Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill




