
ORGANIC CHEMISTRY (LL)-W/MOD.MASTERING.
8th Edition
ISBN: 9780134466712
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 28.5, Problem 13P
- a. Draw the product of the following reaction:
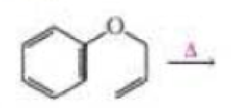
- b. If the terminal sp2 carbon of the substituent attached to the benzene ring is labeled with 14C, where will the label be in the product?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the product of the following reaction. If the terminal sp2 carbon of the substituent attached to the benzene ring is labeled with 14C, where will the label be in the product?
2.
Draw the reaction and resulting product when 1-heptyne is trrated with excess HBr?
Draw the reaction and resulting product when 1-ethyne in H20 is treated with H2SO4 + HgSO4?
Ll.26.
which of the following are carbocations formed during the SN1 reactions shown below?
Chapter 28 Solutions
ORGANIC CHEMISTRY (LL)-W/MOD.MASTERING.
Ch. 28.1 - Prob. 1PCh. 28.2 - Prob. 2PCh. 28.2 - Prob. 3PCh. 28.2 - Give a molecular orbital description for each of...Ch. 28.3 - Prob. 5PCh. 28.3 - Prob. 6PCh. 28.3 - Prob. 7PCh. 28.3 - Prob. 8PCh. 28.4 - Prob. 10PCh. 28.4 - Prob. 11P
Ch. 28.5 - Prob. 12PCh. 28.5 - a. Draw the product of the following reaction: b....Ch. 28.5 - Prob. 14PCh. 28.5 - Prob. 15PCh. 28.5 - Prob. 17PCh. 28.5 - Prob. 18PCh. 28.6 - Prob. 19PCh. 28.6 - Explain why the hydrogen and the methyl...Ch. 28.6 - Chorismate mutase is an enzyme that promotes a...Ch. 28.7 - Convince yourself that the TE-AC method for...Ch. 28 - Draw the product of each of the following...Ch. 28 - Draw the product of each of the following...Ch. 28 - Prob. 25PCh. 28 - Show how norbornance can be prepared from...Ch. 28 - Prob. 27PCh. 28 - Prob. 28PCh. 28 - Draw the product of each of the following...Ch. 28 - Prob. 30PCh. 28 - Prob. 31PCh. 28 - Prob. 32PCh. 28 - Prob. 33PCh. 28 - When the following compound is heated, a product...Ch. 28 - Prob. 35PCh. 28 - Propose a mechanism for the following reaction:Ch. 28 - Prob. 37PCh. 28 - Prob. 38PCh. 28 - Prob. 39PCh. 28 - Prob. 40PCh. 28 - If isomer A is heated to about 100 C, a mixture of...Ch. 28 - Propose a mechanism for the following reaction:Ch. 28 - Prob. 43PCh. 28 - A student found that heating any one of the...Ch. 28 - Prob. 45PCh. 28 - Prob. 46PCh. 28 - Prob. 47P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A hydrocarbon of unknown structure has the formula C8H10. On catalytic hydrogenation over the Lindlar catalyst, 1 equivalent of Η2 is absorbed. On hydrogenation overa palladium catalyst, 3 equivalents of Η2 are absorbed. (a) How many degrees of unsaturation are present in the unknown structure? (b) How many triple bonds are Present? (c) How many double bonds are present? (d) How many rings are present? (e) Draw a structure that fits the data.arrow_forwardA. Draw the structure(s) of the major product(s) from addition of 1 equivalent of HX to the conjugated diene. B. Show the correct mechanism using curved arrowsarrow_forwardWhat stereoisomers are obtained when (S)-3-methyl-1-pentene reacts with Cl2? Draw the structures of the stereoisomers indicating their R/S configuration.arrow_forward
- what is the product of 1-Hexyne reacting with 1 equivalent of HBr? Include E/Z if necessary.arrow_forwardIdentify the correct ranking of the following molecules from most prone to nucleophilic attack at a sp^2 carbon to least. The correct answer is D, but please explain.arrow_forward56. If a trans alkene is reacted with OsO4 in H2S, the product is: Meso compound Hoffman product Markovnikov product Racemic mixturearrow_forward
- What are products formed from the below reaction?Draw the stereoisomers and name themarrow_forwardDraw the products 1. (S)-2-chlorobutane and sodium acetate in DMSO 2. 1-bromopropane and methylamine in acetonitrile.arrow_forwarda.What product(s) (excluding stereoisomers) are formed when Y is heated with Cl2? b.What product(s) (excluding stereoisomers) are formed when Y is heated with Br2? c.What steps are needed to convert Y to the alkene Z?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License