
(a)
Interpretation: The product of given reaction has to be drawn.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or brocken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
There are mainly three types of pericyclic reactions,
- 1) Electrocyclic reactions
- 2) Cycloaddition reactions
- 3) Sigmatropic reactions
In an electrocyclic reaction “one new sigma- bond is formed or brocken.”
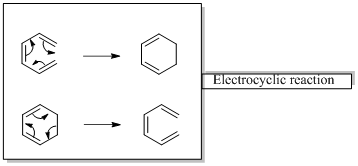
Woodward –Hoffmann rules are the set of rules used to vindicate or predict certain aspects of the stereo chemical outcome and activation energy of pericyclic reactions.
Woodward – Hoffmann rules for Electrocyclic reactions are listed below
A photochemical reaction takes place when a reactant absorbs light and a thermal reaction takes place without the absorption of light.
Woodward – Hoffmann rules for the configuration of electrocyclic reactions are,
(b)
Interpretation: The product of given reaction has to be drawn.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or brocken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
There are mainly three types of pericyclic reactions,
- 1) Electrocyclic reactions
- 2) Cycloaddition reactions
- 3) Sigmatropic reactions
In an electrocyclic reaction “one new sigma- bond is formed or brocken.”

Woodward –Hoffmann rules are the set of rules used to vindicate or predict certain aspects of the stereo chemical outcome and activation energy of pericyclic reactions.
Woodward – Hoffmann rules for Electrocyclic reactions are listed below
A photochemical reaction takes place when a reactant absorbs light and a thermal reaction takes place without the absorption of light.
Woodward – Hoffmann rules for the configuration of electrocyclic reactions are,
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
ORGANIC CHEMISTRY (LL)-W/MOD.MASTERING.
- For the following starting material A, draw the products B and C that you expect to form under the following reaction conditions.arrow_forwardA. Provide the necessary reagents. B. Provide the necessary eragnets and the step by step mechanism.arrow_forwardWrite reasons for the regioselectivity of hydroboration-oxidation of alkene by drawing the reaction and transition states. Simply saying ‘because of Markovnikov’s or anti-Markovnikov’s rule’ is not a correct answer.arrow_forward
- Provide the neccessary reagents next to the arrowsarrow_forwardConsider the conversion of alkyl halide A to ether B. a.Classify the conversion of A to B as substitution, elimination, or addition. b.The reaction rate depends on the concentration of A only. Write the rate equation for the reaction, and explain why the reaction mechanism must involve more than one step. c.Heterolysis of the polar bond in A forms a resonance-stabilized intermediate. Draw all reasonable resonance structures for this intermediate.arrow_forwardDraw structure of substitution product(s) & major elimination productarrow_forward
- Rationalize the selectivity observed in the benzil reduction. Draw the transition states of the nucleophilic attack leading to either meso or racemic products and indicate why one is more stable than the other.arrow_forwarda. What are all the monochlorination product of the reaction with the alkylhalideabove? For each product calculate their relative percentages for the reaction b. What is the Major product formed by the monobromination product of thereaction? Draw the arrow pushing mechanism for the initiation, propagation andtermination steps.arrow_forwardDraw the structure (including stereochemistry) of an alkyl chloride that forms each alkene as the exclusive E2 elimination product.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
