
Concept explainers
Interpretation:
[1,3]sigmatropic migrations of hydrogen cannot occur under thermal conditions but [1,3] sigmatropic migration of carbon can. Reason for this should be explained.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or brocken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
There are mainly three types of pericyclic reactions,
- 1) Electrocyclic reactions
- 2) Cycloaddition reactions
- 3) Sigmatropic reactions
In a sigmatropic reaction “ one new sigma-bond is formed as another breaks.”
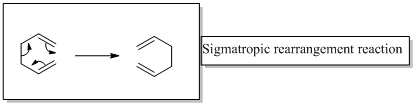
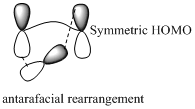
Sigmatropic rearrangement reactions are named with digits. For example a [1, 3] sigmatropic rearrangement describe a reaction in which the residue migrates from position 1 to position 3. Migration of carbon and hydrogen will occur in a sigmatropic rearrangement reaction. When hydrogen migrates in a sigmatropic rearrangement, the s orbital of the hydrogen is partially bonded to both the migration origin and the migration terminus in the transition state. Migration of hydrogen in suprafacial and antarafacial rearrangement can be represented as follows,


Migration of carbon occurs through two ways because it has a two lobed p orbital. Carbon can simultaneously interact with the migration origin and the migration terminus using one lobe of its p orbital.
Migration of carbon in suprafacial and antarafacial rearrangement can be represented as follows,
Carbon migrating with one lobe of its p orbital interacting


Carbon migrating with both lobe of its p orbital interacting

Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
MODIFIED MASTERING CHEMISTRY W/ ETEXT
- The dipole moment of bromobenzene is 5.17 × 10−30 C m and its polarizability volume is approximately 1.5 × 10−29 m3. Estimate its relative permittivity at 25 °C, when its mass density is 1491 kg m−3.arrow_forwardCalculate the relative intensities M+1 and M+2 for CH3COOHarrow_forwardIs structure 4 or 2 the correct option due to sn2 inversion ?arrow_forward
- suggest with a valid reason whether [CoBr6]4- will undergo Jahn-Teller distortionarrow_forwardDoes the exclusion rule apply to H2O?arrow_forward1. List 2 advantages of infrared spectroscopy 2. Explain in detail the work principle of Infrared spectroscopy 3. How many vibrational; modes are there in tetrahedral CH4 molecule 4. Explain out plane and Asymmetrical vibration 5.Explain the principle of operation of UV-Spectroscopy 6. Discuss 2 factors that result in deviation from Beer's lawarrow_forward
- Which has a lower characteristic stretching Frequency, the C=O bond or band C-O bond.arrow_forwardExplain why a molecule with no dipole moment is microwave inactive but may show an infrared spectrum.Which of the following molecules may show infrared absorption spectra and, in each case, briefly explain why this is so: (i) H2, (ii) HCl, (iii) CO2 , and (iv) H2O?arrow_forwardWhat is the Molecular geormetry for SiO32- ?arrow_forward
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning


