
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 28.SE, Problem 17MP
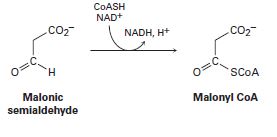
The final step in the
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Propose a mechanism for the TPP-dependent step in isoleucine biosynthesis. Draw the predicted product of this reaction.
Pyruvate carboxylase is thought to activate CO2 by ATP, through formation of carboxyphosphate as an intermediate. Propose a mechanism for the formation of this intermediate.
Propose a mechanism for the conversion of F to toremifene
Chapter 28 Solutions
Organic Chemistry
Ch. 28.1 - Draw the full structure of the DNA dinucleotide...Ch. 28.1 - Draw the full structure of the RNA dinucleotide...Ch. 28.2 - What sequence of bases on one strand of DNA is...Ch. 28.4 - Show how uracil can form strong hydrogen bonds to...Ch. 28.4 - What RNA base sequence is complementary to the...Ch. 28.4 - From what DNA base sequence was the following RNA...Ch. 28.5 - List codon sequences for the following amino...Ch. 28.5 - List anticodon sequences on the tRNAs carrying the...Ch. 28.5 - What amino acid sequence is coded by the following...Ch. 28.5 - What is the base sequence in the original DNA...
Ch. 28.7 - Prob. 11PCh. 28.7 - Prob. 12PCh. 28.SE - Identify the following bases, and tell whether...Ch. 28.SE - Identify the following nucleotide, and tell how it...Ch. 28.SE - Amine bases in nucleic acids can react with...Ch. 28.SE - The final step in DNA synthesis is deprotection by...Ch. 28.SE - The final step in the metabolic degradation of...Ch. 28.SE - One of the steps in the biosynthesis of a...Ch. 28.SE - One of the steps in the metabolic degradation of...Ch. 28.SE - One of the steps in the biosynthesis of uridine...Ch. 28.SE - Human brain natriuretic peptide (BNP) is a small...Ch. 28.SE - Human and horse insulin both have two polypeptide...Ch. 28.SE - The DNA of sea urchins contains about 32% A. What...Ch. 28.SE - The codon UAA stops protein synthesis. Why does...Ch. 28.SE - Which of the following base sequences would most...Ch. 28.SE - For what amino acids do the following...Ch. 28.SE - Prob. 27APCh. 28.SE - Prob. 28APCh. 28.SE - Draw the complete structure of the ribonucleotide...Ch. 28.SE - Draw the complete structure of the...Ch. 28.SE - Give an mRNA sequence that will code for the...Ch. 28.SE - Give an mRNA sequence that will code for the...Ch. 28.SE - What amino acid sequence is coded for by the...Ch. 28.SE - What amino acid sequence is coded for by the...Ch. 28.SE - Prob. 35APCh. 28.SE - Show the steps involved in a laboratory synthesis...Ch. 28.SE - Draw the structure of cyclic adenosine...Ch. 28.SE - Prob. 38AP
Additional Science Textbook Solutions
Find more solutions based on key concepts
For Practice 1.1
Is each change physical or chemical? Which kind of property (chemical or physical) is demonst...
Principles of Chemistry: A Molecular Approach (3rd Edition)
Write a Lewis formula for each of the following organic molecules: C2H3Cl (vinyl chloride: starting material fo...
Organic Chemistry - Standalone book
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living by Chemistry
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
The active ingredient in Tylenol and a host of other over-the-counter pain relievers is acetaminophen (C8H9NO2)...
Chemistry: Atoms First
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (14th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- One of the steps in the metabolic degradation of guanine is hydrolysis to give xanthine. Propose a mechanism.arrow_forwardIn step 7 of fatty-acid biosynthesis (Figure 29-5), dehydration of a -hydroxy thioester occurs to give trans-crotonyl ACP. Is the dehydration a syn elimination or an anti elimination?arrow_forwardAssume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material for the biosynthesis of mevalonate, as shown in Figure 27-7. At what positions in mevalonate would the isotopic label appear?arrow_forward
- In bacteria, much of the putrescine is synthesized, not from ornithine but from arginine, which decarboxylates to yield agmatine. Formulate a plausible pathway from arginine to putrescine, using this intermediate.arrow_forwardAn experimenter using acetyl-CoA with a 14C on the carbonyl group traces the radioactive label in cells synthesizing cholesterol. On what atoms of mevalonate will the label appear?arrow_forwardIs the statement, squalene undergoes cyclization during cholesterol biosynthesis correct?arrow_forward
- The vitamin Niacin is used to form nicotinamide adenosine dinucleotide, which readily shuttles between its oxidized (NAD+) and reduced (NADH) forms. The latter serves as a cellular equivalent to NaBH4. The essential portions of the structures are shown below. Outline a mechanism for the cellular conversion of pyruvate to lactate. (Note: like NaBH4, NADH cannot reduce carboxylic acid carbonyls).arrow_forwardCells can also convert a-keto acids into amino acids, but because the reagents organic chemists use for this reaction are not available in cells, they carry out this reaction by a different mechanism. a. What amino acid is obtained from the reductive amination of each of the following metabolic intermediates in a cell by reductive amination? b. What amino acids are obtained from the same metabolic intermediates when the amino acids are synthesized in the laboratory?arrow_forwardPropose a mechanism for the conversion of (S)-citronellal to isopulegol.arrow_forward
- Which part of the lipoamide is the active part aiding in the chemistry of E2 of the pyruvate dehydrogenase? a) the amino nitrogen of lysine b) the amide nitrogen c) the hydrocarbon d) the carbontl carbon of ltsine e) the disulfidearrow_forwardFADH2 reduces α,β-unsaturated thioesters to saturated thioesters. The reaction is thought to take place by a mechanism that involves radicals. Propose a mechanism for this reaction.arrow_forward(i) Acetylation of glucose with acetic anhydride gives glucosepentaacetate. Write the structure of the pentaacetate.(ii) Explain why glucose pentaacetate does not react with hydroxylamine?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License